
Introduction
Most cosmetic founders spend months chasing the right manufacturer — only to realise they chose the wrong model entirely. From indie startups to established FMCG players, brands routinely outsource production to specialists, but many enter without understanding how third-party manufacturing actually works.
Approximately 60% of cosmetic companies globally now outsource production, with indie brands accounting for roughly 70% of new outsourcing contracts.
The difference between choosing a third-party manufacturer, a contract manufacturer, or a private label provider can determine whether your first product launch costs ₹12 lakh or ₹1.2 crore, and whether your formula remains yours.
This guide is for startups launching their first product, existing brands expanding SKUs, and D2C brands exploring manufacturing partnerships. You'll learn how to choose the right manufacturing model, protect your IP at scale, evaluate certifications that matter, and avoid the costly mistakes that trip up first-time brand founders.
TLDR
- Third-party cosmetic manufacturing outsources formulation, testing, and production to a specialist who manufactures finished products under your brand
- Key model differences: third-party (manufacturer controls formula), contract (you control formula), private label (off-the-shelf formulas)
- Eliminates the heavy capital expenditure of building your own manufacturing facility — freeing budget for brand and product development
- Evaluate manufacturers on GMP certification, IP protection practices, batch consistency, and production capacity — not price alone
- Common pitfalls: skipping stability testing, ignoring documentation requirements, failing to confirm maximum capacity before signing
What Is Third-Party Cosmetic Manufacturing?
Third-party cosmetic manufacturing is a business arrangement in which a brand hires a specialized manufacturer to produce cosmetic products on its behalf. The manufacturer handles production operations (formulation, raw material sourcing, quality testing, packaging) while the brand owns the product concept, branding, and go-to-market strategy.
Who uses this model:
- Beauty startups with limited capital looking to avoid heavy facility investment
- Established brands launching new product categories without disrupting existing production lines
- Influencer-led brands prioritizing speed-to-market over in-house capabilities
- Exporters manufacturing in one country to sell in another
The global personal care contract manufacturing market was valued at $24.18 billion in 2025 and is projected to reach $45.44 billion by 2033, growing at 8.4% CAGR. Asia Pacific dominates with 38.6% market share, while skin care accounts for 36.2% of product segments.
Division of responsibilities:
| What the Manufacturer Handles | What the Brand Retains |
|---|---|
| Raw material sourcing | Brand identity & positioning |
| Formulation (or production to spec) | Marketing strategy |
| Quality testing & documentation | Go-to-market execution |
| Packaging & labeling | Intellectual property over proprietary formulas |
The brand retains full intellectual property over its proprietary formulas, provided this is explicitly defined and protected through non-disclosure and exclusivity agreements.
Third-Party vs. Contract vs. Private Label Manufacturing
What Each Model Actually Means
These three terms get used interchangeably, but they describe fundamentally different relationships between a brand and its manufacturer.
Third-party manufacturing: The brand provides a product brief; the manufacturer handles everything — formulation, ingredient sourcing, and finished production. No in-house R&D required.
Contract manufacturing: The brand owns the formula and specifies every raw material. The manufacturer contributes production capacity only — mixing, filling, and packaging to the brand's exact instructions.
Private label: The brand picks from the manufacturer's existing formulas and applies its own branding. Fastest path to market, but the formula isn't exclusive.
When Each Model Fits
Third-party manufacturing suits:
- Startups without in-house R&D who need full-service support
- Brands requiring custom formulations but lacking formulation expertise
- Companies seeking a balance between customization and operational ease
Contract manufacturing suits:
- Established brands with proprietary formulas needing scaled production
- Companies with in-house chemists who control formulation IP
- Brands requiring maximum control over ingredient quality and sourcing
Private label suits:
- Brands seeking fast, low-investment market entry
- Companies testing product-market fit before investing in custom formulation
- Brands prioritizing speed over product uniqueness
Cost, Control, and Lead Time
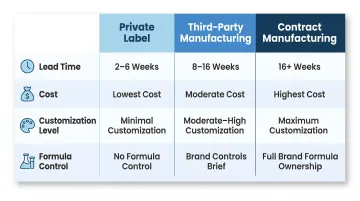
| Model | Typical Lead Time | Cost Level | Customization | Control Over Formula |
|---|---|---|---|---|
| Private Label | 2–6 weeks | Lowest | Minimal | None — formula sold to competitors |
| Third-Party | 8–16 weeks | Moderate | Moderate–High | Brand controls brief; manufacturer controls inputs |
| Contract | 16+ weeks | Highest | Maximum | Full — brand owns and manages formula |
IP Ownership Implications
In private label, the formula belongs to the manufacturer — and can be sold to any competitor willing to put their name on it. In third-party and contract manufacturing, IP ownership must be explicitly defined through:
- Non-disclosure agreements (NDAs)
- Exclusivity clauses preventing formula resale
- Written IP assignment agreements
- Data security and process fragmentation protocols
| Model | Formula Ownership | Ingredient Sourcing | Best Fit |
|---|---|---|---|
| Private Label | Manufacturer | Manufacturer | Fast market entry, testing |
| Third-Party | Brand (if contracted) | Manufacturer | Startups, brands without R&D |
| Contract | Brand | Brand | Established brands, proprietary formulas |
Key Benefits of Third-Party Cosmetic Manufacturing
Reduced Capital Expenditure
Building a GMP-certified cosmetic manufacturing facility requires massive upfront investment. In the US, total CAPEX for a small-scale facility is approximately $510,000, including:
- Manufacturing equipment: $250,000 (mixing, filling, packaging lines)
- R&D and lab setup: $125,000
- Facility and infrastructure: $90,000
- IT and office setup: $45,000
Add $25,000 in monthly fixed operating expenses (rent, utilities, compliance) before accounting for salaries. Break-even typically takes 14 months.
In India, a basic in-house facility requires ₹2-5 crore ($240,000-$600,000), with R&D labs costing ₹50 lakh to ₹1 crore ($60,000-$120,000).
Third-party manufacturing converts these fixed costs into variable ones. You pay only for what you produce, avoiding heavy capital investment in plants, equipment, and quality systems.
Faster Speed-to-Market
Validated production lines, tested raw material suppliers, and regulatory documentation frameworks already exist at established manufacturers. This compresses time from concept to commercial launch.
Timeline comparison:
- Outsourced: Concept to sampling in 4-6 weeks
- In-house: Same process takes 3-4 months even with dedicated resources
Total production lead time (order confirmation to shipment) typically runs 6-10 weeks. High-performing brands reduce lead times by 15-25% through vendor consolidation and standardized components.
In trend-driven categories — serums, sun care, K-beauty-inspired formats — that 6-8 week difference between in-house and outsourced can determine whether a brand captures a trend cycle or misses it entirely.
Access to Specialized R&D Expertise
Top-tier manufacturers employ chemists, dermatologists, and formulation scientists with backgrounds at global ingredient and personal care companies. Most brands cannot afford to hire this expertise independently.
Cost benchmarks:
- Average US cosmetic chemist salary: $86,590 per year
- Cosmetic formulation chemist: $76,409 per year
Third-party manufacturers bring years of application-specific chemistry knowledge that directly affects product outcomes:
- Deliver measurably better performance and shelf stability
- Resolve ingredient compatibility issues before they reach production
- Apply advanced delivery systems — encapsulation, controlled release — that most in-house teams lack access to
- Reformulate quickly when raw material supply or regulations change
For example, Distil's R&D team includes PhDs, MScs, and engineers with 45+ years combined experience from Dow, BASF, L'Oréal, Huntsman, and Reliance Industries. This expertise enables development of high-purity cosmetic esters, tailored UV filters, and mild surfactants for clean-beauty formulations.
Scalability Without Capital Risk
A well-structured partnership allows brands to start at pilot batch volumes and scale to commercial quantities without additional capital investment. The manufacturer absorbs capacity expansion through their facility or partner network.
Before committing to a partner, confirm:
- Maximum batch size and production frequency at full scale
- Lead time changes as volumes increase
- Whether capacity is in-house or relies on subcontractors
A successful launch can stall fast if the manufacturer hits a capacity ceiling — and that conversation is far harder to have after purchase orders are already in.
Regulatory and Compliance Support
Cosmetic regulations vary significantly across markets:
- India: BIS standards, CDSCO manufacturing licenses
- United States: FDA registration, MoCRA compliance
- European Union: EC 1223/2009, mandatory CPSR, CPNP notification
Experienced manufacturers maintain required certifications and generate the documentation brands need for market access:
- Certificates of Analysis (COA)
- Material Safety Data Sheets (MSDS)
- Stability test reports
- Product Information Files (PIF) for EU markets
- Safety assessments and toxicological profiles
How the Third-Party Manufacturing Process Works
Briefing and Product Consultation
The brand shares its product concept with the manufacturer, including:
- Target consumer and positioning
- Performance claims and efficacy goals
- Preferred ingredients or restrictions (vegan, fragrance-free, organic, halal)
- Desired pack format and size
- Target price point and margin requirements
The manufacturer's team uses this brief to propose formulation directions and assess technical feasibility. This stage surfaces potential challenges early: ingredient incompatibilities, stability concerns, and regulatory constraints.
Formulation Development and Sampling
The manufacturer's R&D team develops prototype formulas, producing small-scale lab samples for brand evaluation on:
- Texture and viscosity
- Scent and color
- Skin feel and absorption
- Claimed efficacy (where testable at lab scale)
Expect 4–12 weeks from brief to first samples, depending on product complexity. Multiple rounds of reformulation are common — budget adequate time before expecting a final approved sample.
Custom surfactant development or advanced delivery systems (liposomal encapsulation, time-release) extend timelines. Simple emulsions or oil blends move faster.
Once a sample is approved, it must clear stability testing and regulatory documentation before bulk production begins.
Stability Testing and Regulatory Documentation
Stability testing:
- Accelerated testing (12 weeks at 40°C predicts 1-year shelf life)
- Real-time testing at intended storage conditions
- Freeze-thaw cycles, centrifugation, vibration, light exposure
- Monitoring physical appearance, viscosity, pH, microbial load
Preservation efficacy (challenge testing):
- Ensures preservative system prevents microbial contamination
- Follows ISO 11930 standard
- Mandatory for products containing water
Both stability and preservation results feed directly into the regulatory documentation package:
Regulatory documentation:
- Safety assessments
- Labeling compliance checks
- COA generation
- CPSR for EU markets (qualitative/quantitative composition, toxicological profile, exposure analysis)
Skipping stability testing to meet a launch deadline creates product liability exposure that no timeline saving justifies. Don't cut this step.

Bulk Production, Quality Control, and Delivery
The manufacturer scales from lab batch to bulk production runs. Quality control checks at multiple stages ensure batch-to-batch consistency:
QC checkpoints:
- Raw material inspection (identity, purity, microbial load)
- In-process checks (pH, viscosity, temperature monitoring)
- Finished goods testing (appearance, stability indicators, preservative efficacy confirmation)
Request and review batch records and QC reports routinely. The manufacturer then packages, labels, and dispatches finished products — either to your warehouse or directly to customers via fulfillment arrangements.
How to Choose the Right Third-Party Cosmetic Manufacturer
Key Evaluation Criteria
1. Certifications
Verify before shortlisting:
- GMP certification (ISO 22716 for cosmetics) — mandatory for EU market access
- Market-specific credentials: FDA registration (US), BIS compliance (India)
- Product-specific certifications: Organic, cruelty-free, halal, vegan (if making those claims)
ISO 22716:2007 covers production, control, storage, and shipment of cosmetic products. In 2013, the FDA updated its GMP guidelines to consider ISO 22716 recommendations. EU Regulation 1223/2009 makes compliance mandatory.
2. Formulation Capability
Ask specifically:
- Background of R&D team (academic credentials, prior employers)
- Can they develop custom formulas or only offer standard bases?
- What product categories do they specialize in?
- Do they have in-house testing capabilities or outsource?
3. IP Protection Practices
Ask directly:
- How do they handle formula confidentiality?
- Do they use NDAs and IP assignment clauses?
- Do they sell similar formulas to multiple clients?
- What technical safeguards prevent formula disclosure (process fragmentation, permission-based access)?
4. Batch Consistency Mechanisms
Critical when working with partner manufacturing networks:
- How do they prevent process drift across batches?
- What unified quality systems are in place?
- Can they provide batch records from previous production runs?
- How do they handle deviations or out-of-spec batches?
5. Communication and Transparency
Assess during initial conversations:
- Responsiveness to inquiries
- Willingness to share factory documentation and certifications
- Whether they provide a dedicated point of contact
- Clarity on timelines, costs, and minimum order quantities
Common Mistakes to Avoid
- Choosing solely on lowest price: Cheap inputs and skipped safety testing create liability risk that outweighs any manufacturing savings. A ₹1.6 lakh stability test can save you from a ₹1.6 crore product recall.
- Skipping stability test data: Don't commit to a formulation without reviewing accelerated and real-time stability data. Separation or microbial contamination at the 6-month mark destroys brand credibility.
- Missing IP terms in writing: Get NDAs, exclusivity clauses, and formula ownership terms in signed contracts before sharing any proprietary concepts. Verbal assurances are not enforceable.
- Not checking maximum production capacity: A successful launch can leave you unable to fulfill demand if your manufacturer caps at 5,000 units per month. Confirm scale-up capability before signing.

The Distil Approach
The criteria above aren't abstract — they map directly to what the right manufacturing partner actually looks like in practice. Distil is built around each of them.
R&D-led formulation: Dedicated team with experience from L'Oréal, BASF, Dow, and Huntsman provides application-specific tuning and trials. Custom formulations are developed to precise performance and sensory requirements.
Asset-light flexible manufacturing: Production scales through 20+ vetted partner facilities across India — no capital expenditure required. Brands pay only for what they produce, with full visibility into batch yields from lab to commercial volumes.
IP protection framework: Full data ownership retained by the customer, secured through control layers, process fragmentation, and permission-based access. Proprietary formulations are protected from disclosure to competing clients.
End-to-end partnership: Single point of contact manages the journey from concept to delivery, with real-time visibility across production and supply chain. Unified quality system eliminates process drift across partner facilities.
For brands moving from concept to commercial scale, this structure reduces risk at every stage — from first formula to final shipment.
Frequently Asked Questions
What is third-party manufacturing for cosmetics?
Third-party cosmetic manufacturing is when a brand hires a specialized manufacturer to produce cosmetic products on its behalf — covering formulation, ingredient sourcing, testing, and packaging — while the brand focuses on marketing, sales, and brand building. The manufacturer handles production; the brand owns the product concept and IP.
How do I find a private-label cosmetics manufacturer?
Brands can find private-label manufacturers through B2B sourcing platforms (BeauteTrade, Tradewheel, Wonnda), industry trade shows (Cosmoprof, in-cosmetics, NYSCC Suppliers Day), and direct outreach to certified manufacturers. Request samples and verify GMP certifications before signing any agreement.
What is the difference between third-party and contract manufacturing?
In third-party manufacturing, the manufacturer handles formulation and ingredient sourcing on behalf of the brand. In contract manufacturing, the brand supplies its own formula and raw material specifications while the manufacturer provides production capacity only.
What certifications should a cosmetic manufacturer have?
At minimum, look for GMP certification (ISO 22716 for cosmetics) and market-specific credentials — FDA registration for US markets, BIS compliance for India, CPNP notification for EU. Additional credentials like organic, cruelty-free, or halal certification are important if your brand makes those claims.
Can I protect my formula when working with a third-party manufacturer?
Yes, but protection depends entirely on the agreements in place. Signed NDAs, clear formula ownership clauses, and manufacturers with process fragmentation protocols — which prevent proprietary formulations from reaching competing clients — are essential safeguards. Confirm all terms contractually before sharing any formulation details.


