
Introduction
A single contaminated batch can trigger a cascading crisis: product recall, consumer safety incident, regulatory enforcement action, and permanent brand damage amplified by social media. In 2024 alone, cosmetics topped the European Commission's Safety Gate alerts at 36% of all product notifications—the highest total on record since 2003—with chemical compliance failures driving 97% of cosmetics alerts. In the U.S., the FDA's analysis of adverse event reports from 2004–2016 revealed spikes driven by hair care failures, including a single brand line that generated over 21,000 consumer complaints. Under the Modernization of Cosmetics Regulation Act (MoCRA), the FDA now holds mandatory recall authority when products pose serious health risks.
Brands scaling across multiple manufacturing partners or sourcing specialty actives from diverse suppliers face the steepest challenge: maintaining consistent quality from batch to batch and stage to stage. Quality control (QC) has moved well beyond back-end verification. It now sits at the center of consumer trust, regulatory compliance, and the operational infrastructure that makes scaling possible without sacrificing consistency.
This guide covers what QC in cosmetics manufacturing entails, the critical stages and testing methods, key compliance frameworks, common pitfalls, and best practices for building a quality-first operation at scale.
TLDR
- QC in cosmetics = systematic checks from raw material intake to final batch release ensuring safety, consistency, and compliance
- Four critical stages: raw material inspection, in-process monitoring, finished product testing, packaging validation
- Essential tests: microbiological, stability, physical/chemical, and efficacy validation
- Regulatory frameworks to know: FDA/MoCRA (US), EU Regulation 1223/2009, India BIS, and ASEAN harmonized guidelines
- Scaling QC requires standardized formulations, qualified suppliers, real-time monitoring, and upstream quality partnerships
What Is Quality Control in Cosmetics Manufacturing?
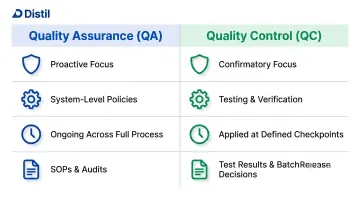
Quality control in cosmetics is the systematic set of procedures and verification checks applied at every production stage to ensure each product meets predefined standards for safety, efficacy, and consistency. QC is distinct from Quality Assurance (QA): QA is the proactive, system-level function that defines policies and standards, while QC is the confirmatory, testing-focused activity that verifies compliance with those standards. The American Society for Quality (ASQ) defines QA as providing confidence that requirements will be fulfilled, and QC as fulfilling those requirements through testing and verification.
QC in cosmetics spans the full production lifecycle: from verifying raw ingredient identity on arrival to testing finished goods before batch release. Products applied directly to skin, hair, and mucous membranes leave no margin for formulation errors or contamination.
The regulatory exposure is real. The European Commission's 2024 Safety Gate report logged over 4,200 enforcement actions against non-compliant cosmetics, and the FDA's MoCRA framework grants mandatory recall authority when responsible persons decline voluntary action. A single QC failure can trigger regulatory suspension, costly recalls, and lasting reputational damage.
| Quality Assurance (QA) | Quality Control (QC) | |
|---|---|---|
| Focus | System-level policies and standards | Testing and verification against those standards |
| Approach | Proactive — prevents non-conformance | Confirmatory — detects non-conformance |
| Timing | Ongoing across the full process | Applied at defined checkpoints |
| Output | Documented procedures, SOPs, audits | Test results, batch release decisions |
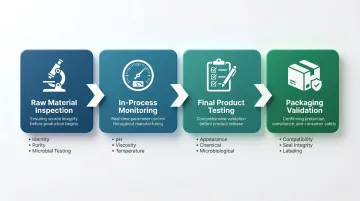
Key Stages of Quality Control in Cosmetics Manufacturing
Raw Material Inspection
QC begins before production starts. Every incoming ingredient must be tested for identity, purity, potency, and microbiological cleanliness. Standard checks include:
- Identity verification using FTIR or NIR spectroscopy
- Certificates of Analysis (CoA) from suppliers
- Microbial count testing (total aerobic, yeast and mold)
- Heavy metal screening: FDA recommends ≤10 ppm lead for lip/externally applied products, <1 ppm mercury as trace impurity, and specific limits for arsenic (≤3 ppm) in color additives
- Pesticide residue testing for botanical and natural ingredients
Supplier qualification runs parallel to incoming inspection. Approved Vendor Lists (AVLs), supplier audits, and multi-source qualification reduce single-source dependency and batch variability.
Brands sourcing from R&D-led specialty chemical suppliers — such as Distil, whose application-specific formulations come with full batch documentation — can cut raw material QC failures before ingredients reach the production floor.
In-Process Monitoring
During formulation and blending, QC teams track critical in-process parameters at defined control points:
- pH, viscosity, temperature
- Homogenization time
- Color and odor consistency
Inline monitoring allows immediate deviation correction rather than retrospective batch rejection — reducing waste and preserving uniformity across runs.
Batch Manufacturing Records (BMRs) and Standard Operating Procedures (SOPs) create the audit trail needed for regulatory compliance, root cause investigation, and batch traceability. This is particularly important for brands operating across multiple manufacturing sites.
Final Product Testing and Packaging Validation
Before any batch is approved, it undergoes comprehensive testing covering:
- Appearance and sensory attributes
- Physical and chemical parameters
- Microbiological safety
No batch is released without meeting 100% of predefined acceptance criteria.
Packaging validation is where many brands lose margin — not in the lab, but in transit or on shelf. Critical checks include:
- Compatibility testing between formulation and packaging material (especially for actives, essential oils, and high-pH formulas)
- Leak-proof sealing verification
- Labeling accuracy checks
- Distribution stress simulation to confirm products arrive intact
For example, China's National Institutes for Food and Drug Control (NIFDC) issued technical guidelines for cosmetic packaging compatibility testing in 2024, requiring accelerated and long-term studies assessing extractables, leachables, and material interactions.
Essential Testing Methods in Cosmetics QC
Microbiological Testing
The full microbiological suite includes:
- Total Aerobic Microbial Count (TAMC)
- Yeast and Mold Count (TYMC)
- Specific pathogen testing: Staphylococcus aureus, Pseudomonas aeruginosa, Candida albicans
- Preservative Efficacy Testing (PET): Also called challenge testing, this confirms the preservation system holds up across the product's claimed shelf life under real-world conditions. ISO 11930 is the reference method.
Acceptance criteria are defined in ISO 17516, which sets microbial limits by product category and requires absence of specified pathogens.
Stability Testing
Stability studies include:
- Accelerated stability: Elevated temperature and humidity (typically 40°C/75% RH)
- Real-time stability: Standard storage conditions over extended periods
Instability indicators to watch:
- Phase separation
- Color change
- Viscosity shift
- pH drift
- Microbial growth
Results inform shelf life labeling, storage recommendations, and packaging decisions. Under MoCRA, FDA does not prescribe specific stability test conditions, but adequate safety substantiation is mandatory — and stability data is typically central to that case.
Physical and Chemical Testing
Main analytical methods include:
- Spectroscopy (IR, UV-Vis): Active ingredient concentration verification
- Chromatography (HPLC, GC): Component separation and contaminant detection
- Rheology: Texture, spreadability, and flow behavior for batch-to-batch sensory consistency
Safety and Efficacy/Claim Validation Testing
Physical and chemical data confirm what's in the formula. Safety and efficacy testing determines whether it's safe to use and whether label claims hold up under scrutiny.
Distinguish between:
- Safety testing: In vitro toxicology, skin irritation, sensitization potential using methods like Human Repeat Insult Patch Test (HRIPT)
- Efficacy testing: Clinical trials with human volunteers, instrumental evaluation (corneometers for hydration, visiometers for wrinkle reduction)
Under MoCRA, cosmetics require "adequate safety substantiation" using scientifically robust methods, though no specific test is mandated. Unsubstantiated claims expose brands to regulatory risk.
Packaging Compatibility Testing
Packaging materials can leach chemicals, trigger oxidation, or react with active ingredients — often without visible signs until after the product reaches the consumer. Compatibility testing evaluates:
- Extractables and leachables: Chemicals migrating from packaging into the formula
- Closure integrity: Seal performance under temperature cycling and transport stress
- Photostability: UV-induced degradation in transparent or semi-transparent containers
- Material reactivity: Interaction between packaging polymers and high-activity ingredients like essential oils, oxidizing agents, or botanicals
Incompatible packaging is a common root cause of stability failures that pass bench testing but fail in the field.
Regulatory Compliance Standards Every Cosmetics Manufacturer Must Know
Cosmetics regulations vary significantly by market — and getting them wrong carries real liability. Here's what applies across the major trading regions.
United States: FDA/MoCRA
The Modernization of Cosmetics Regulation Act (MoCRA) introduced in 2022 requires:
- Facility registration with FDA; renewal every 2 years
- Product listing with ingredient disclosures; annual updates
- Serious adverse event reporting within 15 business days
- Mandatory recall authority: FDA can order recalls if responsible persons refuse voluntary action and serious risk criteria are met
- GMP rulemaking: FDA must establish cosmetic GMP regulations
While the U.S. does not pre-approve cosmetics, manufacturers bear full liability for consumer safety.
European Union: Regulation (EC) No. 1223/2009
The EU cosmetics framework is among the world's most stringent:
- Safety assessment before market entry
- Responsible Person designated within the EU
- Product Information File (PIF) maintained for each product
- Notification via Cosmetic Products Notification Portal (CPNP)
- Strict ingredient prohibitions and animal testing ban
Many brands exporting to Asia, Latin America, or the Gulf use the EU framework as their baseline formulation standard.
India: BIS and CDSCO Standards
Cosmetics are regulated under the Drugs and Cosmetics Act, 1940 and Cosmetics Rules, 2020, administered by CDSCO and state authorities. BIS standard IS 4011:1997 covers methods of test for safety evaluation of cosmetics.
ASEAN: Harmonized Cosmetic Directive
The ASEAN Cosmetic Directive (ACD) harmonizes requirements via a notification system, GMP guidelines, ingredient annexes for prohibited/restricted substances, and adverse event reporting guidance.
Japan: PMD Act and Standards for Cosmetics
Japan's Pharmaceutical and Medical Device Act (PMD Act) distinguishes cosmetics and quasi-drugs. Compliance relies on negative and positive ingredient lists, with stricter pre-market approval for quasi-drugs. Good Quality Practice (GQP) and Good Vigilance Practice (GVP) apply to marketing license holders.
For brands targeting multiple markets, map your formulations against the strictest applicable standard first — it simplifies compliance across the board rather than requiring market-by-market reformulation.

Common QC Challenges and How to Address Them
Ingredient Variability in Natural and Specialty Raw Materials
Natural ingredients—botanical extracts, plant-derived oils, fermentation-based actives—vary in composition by source, season, and harvest lot. Address this by:
- Setting tight specification ranges for each ingredient
- Requiring updated CoAs with every delivery
- Running independent incoming acceptance testing, not relying solely on supplier certification
Batch Consistency Across Distributed Manufacturing Networks
When production is spread across multiple contract manufacturers or sites, maintaining uniform quality is the single hardest challenge. The European Commission's 2024 Safety Gate alerts show that chemical compliance failures—often rooted in inconsistent formulation control—drove the majority of cosmetics notifications. Solutions include:
- Unified quality management system with standardized SOPs
- Shared specification sheets and centralized batch data access
- Defined escalation process for deviations
Distil addresses this directly: its unified quality system standardizes SOPs and batch data access across a 20+ manufacturing partner network, preventing the process drift that typically emerges when production is fragmented across sites.
Keeping Pace with Evolving Regulatory Requirements
Cosmetics regulations in the U.S., EU, and major emerging markets shift regularly—and brands that treat compliance reactively face costly reformulations and market re-entry delays. Make regulatory monitoring an ongoing function, not a one-time audit. Practical steps include:
- Assigning a dedicated team member or external partner to track regulatory updates across key markets
- Maintaining a live ingredient risk register flagging restricted or watch-listed substances
- Building reformulation lead times into product development cycles before regulatory deadlines hit
Best Practices for Building a Quality-First Cosmetics Manufacturing Process
Standardize Formulations and Document Everything Rigorously
SOPs, BMRs, deviation logs, and calibration records are the foundation of reproducible quality. Standardize:
- Ingredient concentrations
- Processing sequences
- Equipment calibration protocols
- Environmental conditions
This ensures every batch is made the same way, regardless of shift, operator, or facility.
Build a Bottom-Up Quality Culture Through Continuous Training
Quality systems fail when QC is treated as a management function rather than a frontline responsibility. Regular training delivered to production workers — not just QC managers — creates an environment where deviations are caught at the source. That training should cover:
- SOP execution and updates
- Safety protocols and handling procedures
- Regulatory requirements relevant to each role
Leverage Real-Time Monitoring and Digital QC Tools
IoT-enabled sensors, automated deviation alerts, and digital checklists continuously track in-process parameters — pH, temperature, viscosity — and flag anomalies the moment they occur. This visibility stops defective batches before they reach packaging and accelerates root cause investigation.

Qualify and Continuously Audit Your Suppliers
Ingredient quality directly determines what you can control in production. Supplier audits, approved vendor lists (AVLs), and multi-source qualification should be recurring processes — not one-time onboarding steps:
- Require updated CoAs with every delivery
- Conduct periodic facility audits
- Evaluate supplier quality performance as a formal KPI
FDA cosmetic GMP guidance and ISO 22716 both emphasize supplier qualification, CoA verification, incoming QC sampling, quarantine, and traceability.
Partner with Specialty Chemical Providers That Embed QC at the Formulation Stage
For brands sourcing specialty chemicals and active ingredients, the quality of upstream inputs directly determines downstream QC outcomes. Distil, for example, delivers application-specific formulations with full batch documentation and real-time batch yield visibility. Its unified quality system spans a network of 20+ manufacturing partners, which means process drift is managed systematically rather than caught after the fact.
Frequently Asked Questions
What is quality control in cosmetics?
Quality control is the systematic process of testing and verifying that every cosmetic product meets defined standards for safety, efficacy, and consistency across each stage of production—from raw material intake to final batch release.
What is raw material quality control in cosmetics manufacturing?
Raw material QC involves verifying the identity, purity, potency, and microbiological status of every incoming ingredient before it enters formulation. Raw material QC involves verifying the identity, purity, potency, and microbiological status of every incoming ingredient before it enters formulation. Common tools include Certificates of Analysis, FTIR testing, microbial counts, and heavy metal screening.
What are the key stages of quality control in cosmetics manufacturing?
The four core stages are: raw material inspection, in-process monitoring during production, finished product testing before batch release, and packaging validation.
What is the role of QA in the cosmetics industry?
Quality Assurance defines and governs the policies, procedures, and standards that maintain product quality at the system level. QC is the hands-on testing activity that confirms compliance with those standards.
What testing methods are used in cosmetics quality control?
The main categories are:
- Microbiological testing: TAMC, TYMC, pathogen testing, preservative efficacy
- Stability testing: Accelerated and real-time studies
- Physical and chemical testing: Spectroscopy, chromatography, rheology
- Efficacy and claim validation: Performance substantiation
- Packaging compatibility: Migration, seal integrity, material interaction
What is GMP in cosmetics manufacturing?
Good Manufacturing Practices are internationally recognized standards governing manufacturing conditions, documentation, personnel training, and quality systems. ISO 22716 is the primary cosmetics GMP standard, with compliance required across most major markets, including the U.S., EU, and India.


