
Introduction
Picture a mid-sized Indian cosmetic brand that spent ₹8 lakh developing a "clean" face cream — only to recall 15,000 units six months post-launch when consumers reported separation, rancid odour, and mould growth. The root cause? The brand skipped accelerated stability testing and launched based on a three-week visual check. This single decision triggered ₹12 lakh in direct losses, negative online reviews, and a regulatory notice from CDSCO.
That outcome was preventable. Stability testing exists to confirm that a formulation maintains its intended safety, appearance, texture, and efficacy — from manufacture through the end of its claimed shelf life, under real-world storage and use conditions.
For brands selling in India, stability data satisfies CDSCO expectations and BIS norms. For those targeting export markets, it's a legal requirement: EU Regulation 1223/2009 mandates documented stability evidence before a product reaches shelves.
This article covers the parameters stability testing evaluates, the two primary testing approaches, regulatory expectations across India, the EU, and the US, and the failure modes that most often derail products post-launch.
TLDR
- Stability testing verifies that a cosmetic product maintains safety, appearance, and performance throughout its shelf life under expected storage conditions
- Testing evaluates physical, chemical, microbiological, and packaging compatibility parameters at defined intervals
- Two primary methods — real-time testing (up to 36 months) and accelerated testing (3-6 months at 40°C/75% RH) — typically run in parallel
- Regulatory requirements vary by market: EU Regulation 1223/2009 mandates stability data in the CPSR, India's BIS and CDSCO frameworks require safety documentation, and US MoCRA holds manufacturers accountable for product safety even without an explicit stability mandate
- 76.8% of FDA cosmetic recalls from 2011–2023 involved microbial contamination, making preservative efficacy testing non-negotiable for any water-containing formulation
What Is Product Stability Testing in Cosmetics and Why Does It Matter?
Stability testing is the structured process of evaluating whether a cosmetic formulation maintains its specified quality, safety, and performance attributes — including appearance, smell, texture, and efficacy — over its intended shelf life under expected storage and use conditions. For manufacturers, it's the primary mechanism for catching failure before it reaches the consumer.
Products that fail in-market due to phase separation, rancidity, loss of active efficacy, or packaging leakage generate returns, negative reviews, and regulatory recalls — and the scale of these failures is significant. According to the European Commission's 2025 Safety Gate Report, cosmetics accounted for 36% of 4,671 safety alerts in 2025 — the highest of any product category — with total alerts up 13% from 2024 and more than double since 2022.
In the US, a cross-sectional study published in the Journal of the American Academy of Dermatology analyzed 334 FDA cosmetic recalls from 2011 to 2023, covering over 77 million units. The overwhelming majority — 76.8% — were due to microbial contamination, primarily Pseudomonas and Burkholderia species. These failures were voluntary and manufacturer-initiated, underscoring that stability testing is fundamentally a manufacturer responsibility.
Regulatory Requirements by Market
What constitutes a mandatory obligation versus a best practice varies significantly depending on where a product is sold:
- EU: Stability data is a legal requirement under Regulation 1223/2009, Annex I, Part A. The Cosmetic Product Safety Report (CPSR) must include documentation of "physical/chemical characteristics and stability of the cosmetic product under reasonably foreseeable storage conditions."
- US: The FDA does not mandate stability testing under the Modernization of Cosmetics Regulation Act (MoCRA, 2022), but manufacturers carry full legal responsibility for product safety. The Personal Care Products Council (PCPC) strongly recommends stability testing as part of safety substantiation.
- India: BIS standards and the Drugs and Cosmetics Rules require manufacturers to document product safety and quality. Brands exporting to the EU or US must generate CPSR-grade stability data and meet GMP requirements.

Regardless of which regulatory framework applies, stability testing is not purely a formulation concern — it starts with ingredient quality. Batch-to-batch inconsistencies in raw materials (moisture content, purity, pH) can unpredictably alter stability outcomes, and variations in natural raw material quality are among the most commonly cited contributors to batch rejection and in-market failure.
Suppliers that enforce batch consistency and application-specific ingredient validation reduce this risk before it reaches the lab. Distil's R&D-managed supply model, for example, uses a unified quality system across its manufacturing partner network to eliminate process drift at the source — a critical upstream control for formulators running accelerated stability programs.
Key Parameters Evaluated in Cosmetic Stability Testing
Stability testing evaluates multiple interdependent parameters to confirm that a product remains safe and effective throughout its shelf life.
Physical and Organoleptic Parameters
These parameters assess consumer-facing attributes:
- Appearance: Phase separation, sedimentation, syneresis (liquid seepage), creaming, or colour change
- Colour: Fading, browning, or inconsistency across batches
- Odour: Development of rancid, off, or unexpected smells
- Viscosity/Rheology: Thinning, thickening, or loss of texture
- Texture: Changes in spreadability, smoothness, or grittiness
Physical instability signals formulation failure directly. A serum that separates or a cream that thins loses perceived value instantly — and consumer trust along with it.
Chemical Parameters
Chemical testing tracks the integrity of actives and preservatives:
- pH monitoring: Shifts in pH can trigger ingredient degradation or skin irritation
- Active ingredient assay: Confirms no degradation of functional ingredients (e.g., vitamin C, retinol)
- Oxidation markers: Peroxide value, rancidity indicators, and detection of aldehyde byproducts
- Preservative concentration: Verifies that antimicrobial agents remain within effective ranges
- Degradation byproduct assessment: Identifies harmful or allergenic breakdown products
Why it matters: Chemical instability directly affects both efficacy claims and safety. A sunscreen with degraded UV filters fails to protect; a product with excessive formaldehyde from preservative breakdown becomes a health hazard.
Microbiological Parameters
Microbial testing safeguards against contamination:
- Bioburden testing: Quantifies total aerobic microbial count (TAMC) and total yeast/mold count (TYMC) per USP <61>
- Pathogen absence verification: Tests for Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Candida albicans, and Salmonella per USP <62>
- Preservative efficacy challenge testing: Per USP <51> or ISO 11930, products are inoculated with challenge organisms and sampled at 7, 14, and 28 days to confirm preservative performance
Regulatory microbial limits:
- Category 1 products (eye area, mucous membranes, children under 3): Maximum 100 CFU/g or mL
- Category 2 products (other topical): Maximum 1,000 CFU/g or mL
Water-rich and natural/organic formulations face elevated microbial risk. According to research published in Molecules (2018), products with water activity above 0.75 support microbial growth. Between 2008 and 2014, 62 cosmetic products were recalled through EU RAPEX specifically due to microbial contamination.
Ingredient quality plays a direct role here. High-purity raw materials with low bioburden profiles and minimal residual catalysts or fatty acids give formulators a more predictable foundation for preservative system design — reducing microbial risk before the challenge test even begins.
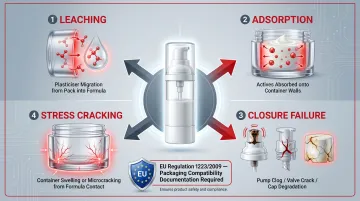
Packaging Compatibility
Packaging is an active participant in product stability:
- Leaching: Migration of plasticisers, stabilisers, or monomers from packaging into the formulation
- Adsorption: Loss of actives or fragrance components onto container surfaces
- Closure integrity: Pump clogging, valve failure, or cap cracking
- Stress cracking and swelling: Physical degradation of containers due to ingredient-pack interaction
- Label/decoration durability: Ink bleed, adhesive failure, or barcode illegibility
Research on mandarin essential oil in HDPE and PET containers documented measurable absorption of d-Limonene and gamma-Terpinene, and migration of the antioxidant Irganox 1076 from HDPE into the formulation. HDPE showed greater absorption than PET, and scanning electron microscopy revealed surface roughening and microcracking after extended contact.
Essential-oil-rich, acidic, or fragrance-forward formulations are particularly prone to container degradation and leaching. EU Regulation 1223/2009, Annex I, requires explicit documentation of "impurities, traces and information about the packaging material" in the CPSR — making packaging compatibility testing a regulatory obligation, not just a quality check.
Accelerated vs. Real-Time Stability Testing: What Manufacturers Need to Know
Cosmetic stability testing employs two complementary approaches to balance speed with accuracy.
Real-Time Stability Testing
Products are stored under typical ambient conditions — 25°C ± 2°C / 60% RH ± 5% RH for Climatic Zones I-II (Europe, North America) or 30°C ± 2°C / 65% RH ± 5% RH for Zones III-IV (India, Southeast Asia) — and monitored at defined intervals:
- Every 3 months in Year 1
- Every 6 months in Year 2
- Annually thereafter
Duration: Typically 24-36 months to cover the full intended shelf life.
Real-time data is the gold standard for setting expiration dates — but waiting 24-36 months to confirm shelf life before launch is rarely feasible on its own.
Accelerated Stability Testing
Products are exposed to elevated temperature and humidity stress — 40°C ± 2°C / 75% RH ± 5% RH per ICH Q1A(R2) — to simulate long-term degradation in a compressed timeframe.
Standard protocol:
- Minimum 3 time points: 0, 3, and 6 months
- Industry practice: 6 months of accelerated data supports a tentative 24-month shelf-life claim while real-time studies continue
This approach delivers fast feedback for launch decisions. Results must still be confirmed by concurrent real-time data before finalizing shelf-life claims.
Supplementary Stress Conditions
Freeze-thaw cycling simulates cold-chain transport and consumer misuse (such as products left in cars):
- Standard protocol: -10°C for 24 hours, then 25°C for 24 hours (3 cycles minimum)
- Rigorous protocol: -10°C to 45°C, 5 cycles for emulsions and serums
Photostability testing (ICH Q1B) applies to transparent packaging and light-sensitive actives like retinol and vitamin C:
- Exposure requirement: ≥ 1.2 million lux hours of visible light and ≥ 200 watt hours/m² of near-UV energy
- Inadequate packaging protection requires either a redesign or added labeling (such as "Store in a dark place")
Centrifugation and vibration testing simulates transport stress and exposes latent emulsion instability or closure defects before products reach market.

Multi-Batch Requirements
Physical stress testing reveals how a formulation behaves — but none of that data holds up if the product itself isn't consistent between batches. For OTC-cosmetic crossover products (sunscreens, anti-acne treatments), the FDA expects stability data from at least 3 primary batches manufactured under the same conditions intended for commercial distribution.
Batch-to-batch consistency — from ingredient sourcing through production — is essential for generating credible stability data. Process drift or supplier variability can invalidate testing efforts. For ingredient suppliers, this means controlling upstream parameters: peroxide value in cosmetic esters, bioburden in essential oils, and particle size in UV filter dispersions must remain within specified limits across every batch used in stability studies.
Regulatory Standards and Compliance Requirements
Global Frameworks
| Framework | Scope | Key Requirements |
|---|---|---|
| EU Regulation 1223/2009 | Cosmetics sold in the EU | CPSR must include stability data under "reasonably foreseeable storage conditions" (Annex I, Part A); mandatory for market access |
| ICH Q1A(R2) | OTC drugs, adapted for cosmetics | Defines long-term, intermediate, and accelerated testing conditions across climatic zones |
| ISO/TR 18811:2018 | Global cosmetics methodology | Guidelines on stability testing (non-mandatory); manufacturer specifies and justifies protocol |
India-Specific Regulatory Landscape
BIS Standards: Schedule S of the Drugs and Cosmetics Rules references 30 Indian Standards (IS 3959 for skin powders, IS 6608 for skin creams, IS 15153 for face packs, etc.) that finished cosmetics must conform to. These standards focus on quality specifications rather than mandating formal stability protocols.
CDSCO Requirements: Cosmetic manufacturers must document product safety and quality. For export to the EU, Indian brands must generate a full CPSR per EU Regulation 1223/2009, including stability data under appropriate climatic conditions.
Climatic Zone IV (India): Long-term storage condition is 30°C ± 2°C / 65% RH ± 5% RH, which is more stringent than Zones I-II (25°C/60% RH). Products stable for the Indian market often exceed EU/US long-term requirements.
US MoCRA and FDA Position
MoCRA (2022) introduced:
- Mandatory GMP requirements
- Adverse event reporting (serious events within 15 business days)
- Mandatory recall authority
- Facility registration and product listing
Despite these provisions, MoCRA stops short of explicitly mandating stability testing. Manufacturers retain full responsibility for product safety, and the FDA treats shelf life determination as "part of the manufacturer's responsibility."
Labelling Decisions Based on Stability Data
| Label Type | Applies When | Requirement |
|---|---|---|
| EU Best-Before Date (Article 19) | Minimum durability < 30 months | Egg timer symbol + date on label |
| Period After Opening (PAO) | Minimum durability ≥ 30 months | Open jar symbol + duration in months or years |
| US Expiration Date | OTC-cosmetic crossover products only | Expiration date required per 21 CFR 211, unless stability data confirms >3 years stability |

The FDA does not require expiration dating or PAO labelling for standard cosmetics. That said, stability data often informs voluntary shelf life claims — and for any product straddling the OTC-drug line, it becomes a compliance requirement.
Common Stability Failures and How to Prevent Them
Most Frequent In-Market Failure Patterns
Emulsions thinning or phase-separating:
- Cause: Emulsifier degradation or temperature cycling during transport
- Prevention: Freeze-thaw cycling during testing; select robust emulsifier systems
Colour cosmetics fading or developing off-odours:
- Cause: Pigment and oil instability; oxidation
- Prevention: Photostability testing; antioxidant addition; peroxide value monitoring
Preservative failure in water-rich or natural formulations:
- Cause: Microbial contamination, inadequate preservative concentration, or loss of efficacy due to pH shift
- Prevention: Preservative efficacy challenge testing (USP <51> or ISO 11930); water activity management
Packaging-related failures:
- Cause: Pump clogging, closure cracking, label bleed, or leaching
- Prevention: Packaging compatibility testing for formula-pack pairs; stress testing closures with essential-oil-rich products
Root Causes Often Overlooked
Ingredient variability between supplier batches:
Different water content, trace impurities, or particle size can alter pH, emulsion stability, or preservative efficacy in ways that pass initial QC but surface as failures during long-term storage.
Solution: Work with a supply chain partner that enforces batch consistency and application-specific ingredient validation. For aroma chemicals like linalool and citral, this means controlled impurity profiles and precise isomer distributions — both of which directly affect whether a fragrance performs consistently across batches.
These root causes share a common thread: decisions made upstream — during sourcing and supplier qualification — determine whether a formulation survives long-term storage or fails in the field.
Prevention Strategies
- Build stability sign-off into the product development gate process: No launch without completed accelerated and real-time testing
- Conduct stability impact assessments whenever formulation, packaging, supplier, or manufacturing site changes occur
- Retain production batch samples for ongoing shelf-life confirmation monitoring post-launch
- Select packaging materials validated for compatibility with the specific formulation type (e.g., amber or UV-blocking materials for light-sensitive actives)
- Source specialty ingredients from suppliers with unified quality systems and low-impurity profiles to minimise downstream stability risk

Frequently Asked Questions
What is stability testing in cosmetics?
Cosmetic stability testing is a structured scientific process to verify that a product maintains its intended safety, appearance, texture, and performance throughout its labelled shelf life under expected storage and use conditions.
How do you test for stability in cosmetics?
Stability testing combines accelerated testing (40°C/75% RH conditions) with real-time testing conducted over months to years at ambient conditions. Both approaches use periodic sampling to evaluate physical, chemical, microbiological parameters, and packaging compatibility.
How long does stability testing take for cosmetics?
Accelerated studies typically run 3-6 months, while real-time studies run for the full duration of the intended shelf life (commonly 24-36 months). Most manufacturers conduct both in parallel to support launch decisions while long-term data is gathered.
What is the QC test for cosmetics?
QC testing in cosmetics covers several distinct assessments, each serving a different quality gatekeeping function:
- Physical and chemical analysis: pH, viscosity, active assay
- Microbiological testing: bioburden counts, pathogen absence
- Preservative efficacy testing
- Stability testing
What happens if a cosmetic product fails stability testing?
Failures require reformulation (ingredient or preservative system changes), packaging redesign, or revision of shelf life claims. Products launched without adequate stability evidence risk in-market recalls, regulatory penalties, and brand damage.
Does ingredient quality affect cosmetic stability testing results?
Yes. Raw material quality and batch-to-batch consistency directly influence stability outcomes. Variations in purity, pH, or moisture content can alter preservative efficacy, emulsion stability, and active ingredient integrity — which is why upstream ingredient quality control is inseparable from reliable stability testing.
Final Takeaway:
Cosmetic stability testing is a scientific and commercial safeguard — one that determines whether a product succeeds or fails in consumers' hands. For Indian brands with export ambitions or quality-conscious domestic positioning, treating ICH-aligned stability testing as the de facto global standard is essential.
The numbers make the case plainly: microbial contamination drives 76.8% of FDA recalls, and the EU mandates stability data as a legal prerequisite for market access. Investing in rigorous stability testing costs a fraction of what a recall, regulatory penalty, or brand crisis would.


