
Introduction
Oil and water don't mix — and in formulation science, that incompatibility is a daily problem. Personal care, food, pharmaceutical, and industrial manufacturers all depend on stable emulsions where oil and water coexist without separating, losing texture, or degrading actives.
An unstable emulsion means wasted R&D investment, compromised product efficacy, and failed batches. Getting the emulsifier selection right is not optional.
Water-in-oil (W/O) emulsifiers solve this problem. These amphiphilic molecules stabilize water droplets within an oil continuous phase, creating emulsions with unique performance characteristics. Selecting the right W/O emulsifier determines product texture, shelf stability, water resistance, and occlusive barrier properties—critical factors for applications ranging from sunscreens to margarine to pharmaceutical ointments.
This article covers the mechanism of action behind W/O emulsifiers, how to select them using HLB and HLD systems, common emulsifier types, formulation stabilization techniques, and cross-industry applications.
TLDR:
- W/O emulsifiers with HLB values of 1-6 stabilize water droplets dispersed in oil continuous phases
- These systems deliver superior occlusivity, water resistance, and TEWL reduction compared to O/W emulsions
- Stabilization requires oil-phase thickeners, electrolytes in the water phase, and precise temperature control
- Applications span sunscreens, barrier creams, margarine, pharmaceutical ointments, and controlled-release drug delivery
- HLD framework offers more precise formulation control than HLB alone for complex systems
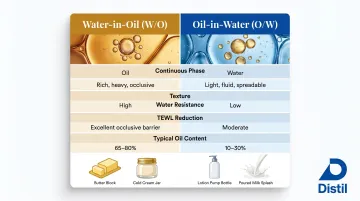
W/O vs O/W Emulsions: Understanding the Core Distinction
The continuous phase determines emulsion type. In oil-in-water (O/W) emulsions, oil droplets disperse in an aqueous continuous phase—think milk or body lotion. In water-in-oil (W/O) emulsions, water droplets disperse in an oil continuous phase—butter and cold cream are classic examples.
This structural difference drives end-product characteristics. W/O emulsions produce heavier, richer textures with higher water resistance—the oil-continuous phase creates a hydrophobic coating that resists washoff and reduces transepidermal water loss (TEWL).
O/W emulsions feel lighter, absorb faster, and spread more easily. The tradeoff: less occlusion and shorter-lasting moisture retention.
W/O vs O/W Comparison:
| Feature | W/O Emulsions | O/W Emulsions |
|---|---|---|
| Continuous Phase | Oil | Water |
| Texture | Heavy, rich, occlusive | Light, fast-absorbing |
| Water Resistance | High (resists washoff) | Low (easily removed) |
| TEWL Reduction | Significant (barrier effect) | Moderate |
| Common Examples | Butter, margarine, cold cream, barrier creams | Milk, lotions, vanishing creams |
| Typical Oil Content | 45-80% | 5-30% |
The barrier advantage of W/O systems is measurable. A 2025 Pharmaceutics review found that formulations with 55% water content maintained occlusive effects for over 5 hours—comparable to petrolatum—while increasing to 70% water (typical of O/W systems) sharply reduced barrier efficacy.
How Water-in-Oil Emulsifiers Work: The Science of Stability
Amphiphilic Molecular Architecture
Water-in-oil emulsifiers are amphiphilic molecules with a hydrophilic (polar) head group and a hydrophobic (nonpolar) tail. In W/O emulsions, molecular orientation reverses compared to O/W systems: nonpolar tails extend outward into the oil continuous phase while polar head groups point into the dispersed water droplet.
When the emulsifier adsorbs at the oil-water interface, it reduces interfacial tension — the energy barrier that normally prevents oil and water from mixing. Lower interfacial tension makes it easier to disperse water into fine droplets and prevents the system from reverting to separated phases.
Interfacial Film Formation
Emulsifiers arrange themselves around each water droplet at the oil-water interface, forming a protective film that prevents coalescence. Two mechanisms are at work, depending on emulsifier type:
- Steric stabilization (non-ionic emulsifiers): Bulky polar head groups physically block droplets from merging, with elastic repulsion forces pushing approaching droplets apart.
- Electrostatic repulsion (ionic emulsifiers): Charged droplet coatings repel each other — though this mechanism loses effectiveness in W/O systems, where the oil phase's low dielectric constant limits ionic interactions. Non-ionic steric stabilization is therefore the dominant mechanism in practice.
The Gibbs-Marangoni Effect
When droplets approach each other, emulsifier molecules are swept from the contact zone, creating an interfacial tension gradient. The resulting flow drives liquid back into the thinning film, pushing droplets apart and preventing coalescence. The interfacial dilational modulus—resistance to deformation—measures how effectively this mechanism operates.
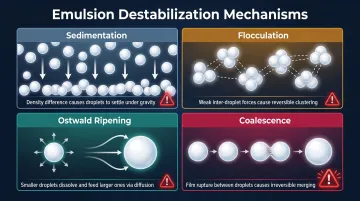
Four Destabilization Pathways
Without emulsifiers — or when they underperform — four mechanisms drive phase separation:
- Sedimentation: Denser water droplets sink through the lighter oil continuous phase (the inverse of creaming in O/W emulsions). The Stokes velocity equation predicts sedimentation rate from droplet size and density difference.
- Flocculation: Droplets aggregate without merging, pulled together by van der Waals attraction. Non-ionic steric barriers suppress this tendency.
- Ostwald ripening: Large droplets grow at the expense of small ones through differential solubility. Dissolved electrolytes in the aqueous phase reduce water solubility in oil, slowing this process.
- Coalescence: Film thinning and rupture between adjacent droplets — the primary failure mode in W/O systems, and the one that robust interfacial films are specifically engineered to prevent.
Types of Water-in-Oil Emulsifiers and Their HLB Values
The HLB Framework for W/O Selection
The Hydrophilic-Lipophilic Balance (HLB) scale runs from 0 to 20, with lower values indicating more oil-soluble (lipophilic) surfactants. According to the Bancroft rule, surfactants with HLB < 7 form W/O emulsions; surfactants with HLB > 7 form O/W emulsions. The practical working range for W/O emulsifiers is approximately HLB 1–6.
Each oil phase has its own required HLB value. Formulators typically use oil-phase HLB charts — or blend two emulsifiers to hit a target HLB — to optimise stability for their specific formulation.
Confirmed W/O Emulsifier HLB Values
| Emulsifier | HLB Value | Typical Use Level | Source |
|---|---|---|---|
| Sorbitan monooleate (Span 80) | 4.3 ± 1.0 | 1–5% | Sigma-Aldrich |
| Glycerol monooleate (GMO) | 3.8 | 2–6% | Mazol Technical Bulletin |
| Lecithin (soy) | 4.0–4.5 | 1–3% | ScienceDirect |
| PGPR (polyglycerol polyricinoleate) | ~1.5–4 | 5% | Food Sci. Biotechnol. |
Span 80's HLB of 4.3 from Sigma-Aldrich is the most precisely documented of these entries.
Bio-Based and Natural W/O Emulsifiers
Green and natural emulsifiers held 72.0% revenue share in the personal care emulsifier market in 2024, valued at USD 565.1 million. This shift reflects consumer preference for clean-label ingredients and stricter regulations in Europe (REACH, EU Cosmetics Regulations).
Natural W/O emulsifier options:
- Polyglycerol esters: Large polar headgroups provide enhanced stability and green credentials
- Lecithin (soy or sunflower): Functions in the HLB 4.0–4.5 range; widely accepted in both food-grade and cosmetic applications


