
Introduction
Many personal care founders struggle with a pivotal operational decision: should we manufacture in-house or outsource production? The capital barrier makes the case plainly: setting up a GMP-compliant cosmetics facility requires a minimum USD 5,10,000 in capital expenditure before you account for working capital, R&D staff, or regulatory infrastructure. For most emerging brands focused on building market presence and customer loyalty, that capex is prohibitive — and a distraction from where early-stage investment actually compounds.
Outsourcing cosmetics manufacturing means delegating production to a third-party specialist while retaining ownership of your formulation, brand identity, and intellectual property. For founders and operators weighing that decision, execution matters as much as the choice itself.
This checklist covers the full process: what to prepare internally, how to evaluate contract manufacturers, what your agreements must include, and which mistakes consistently derail early outsourcing relationships.
TL;DR
- Outsourcing means contracting a third party to produce your formulations, freeing you to focus on brand and growth
- Finalize your formulation brief, MOQ expectations, target market compliance, and certifications before engaging partners
- Evaluate partners on R&D capability, GMP certifications, IP protection practices, and quality systems — price is the last thing to optimize
- Lock down formulation ownership, benchmark samples, batch codes, and defect escalation protocols in your contract before production begins
- Common mistakes: insufficient IP protection, vague briefs, skipping benchmark samples, and undefined defect escalation
What Is Outsourcing Cosmetics Manufacturing?
Outsourcing cosmetics manufacturing involves engaging a contract manufacturer or specialty chemicals platform to produce finished goods based on your specifications: formulation development, blending, filling, and packaging. The manufacturer provides the production infrastructure and expertise while you retain control of the brand, IP, and go-to-market strategy.
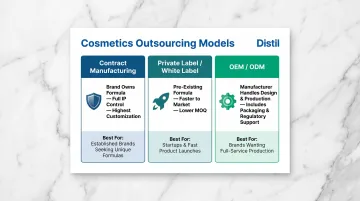
Three Main Outsourcing Models
Contract Manufacturing:
- You own the formula; the manufacturer executes production to your exact spec
- Best for brands needing differentiation and full IP control
Private Label / White Label:
- Faster to market with lower MOQs, but less product differentiation
- Best for market testing or cost-sensitive launches
OEM/ODM:
- Manufacturer handles design and production; often includes packaging and regulatory support
- Best for brands outsourcing both formulation and production decisions
These terms are often used interchangeably, which creates real confusion. The model you choose directly shapes your IP ownership, cost structure, and speed to market — so getting this right matters before you shortlist a single manufacturer.
Why Cosmetic Brands Outsource Manufacturing
Building an in-house facility requires substantial capital expenditure (capex), skilled R&D headcount, GMP-compliant infrastructure, and regulatory expertise — none of which are core functions for a brand focused on marketing, distribution, and customer experience.
Approximately 70% of beauty and personal care brands worldwide outsource production, and the global personal care contract manufacturing market is projected to reach USD 45.44 billion by 2033 at a CAGR of 8.4%.
What Outsourcing Enables at Early-to-Mid Scale
- Access to specialized formulation knowledge without hiring full-time PhDs
- Faster iteration cycles through established R&D pipelines
- Flexible batch scaling without fixed production overhead
- Established supplier networks for raw materials and packaging
In India, in-house manufacturing setup costs INR 50 lakhs to 2+ crores, while outsourcing setup requires only INR 10-15 lakhs excluding launch costs. Brands typically need INR 3-5 crores in annual revenue before in-house manufacturing becomes economically viable.
Compliance and Quality Benefits
Established contract manufacturers already hold ISO 22716 (GMP for cosmetics) certifications and regulatory experience across markets. Under the FDA's Modernization of Cosmetics Regulation Act (MoCRA), manufacturers must register facilities biennially and report serious adverse events within 15 business days — a compliance burden that contract manufacturers absorb across dozens of brand clients.
As of March 2026, 15,309 facilities and 1,102,092 product listings are registered under MoCRA. Outsourcing transfers that administrative weight to your contract manufacturer, reducing your risk of recalls or market entry delays.
The Complete Checklist: How to Outsource Cosmetics Manufacturing
This is an 8-step operational checklist that walks through the process sequentially — from internal preparation to post-launch quality management.
Step 1: Finalize Your Product Brief and Formulation Requirements
The first step is internal: document exactly what you need the manufacturer to produce.
Include in your brief:
- Formulation type (emulsion, serum, anhydrous, powder)
- Target skin/hair concern and desired performance outcomes
- Preferred ingredient inclusions (actives, naturals, certifications)
- Ingredient exclusions and banned substances for your target market
- Preferred texture, fragrance profile, color, and viscosity
- Packaging format and fill weight
The more specific the brief, the fewer revision cycles required. If formulation development is needed (not just filling a finalized formula), specify performance benchmarks and certifications the formula must meet — organic, vegan, halal, cruelty-free.
Step 2: Define Batch Size, MOQ, and Scale Expectations
MOQ varies dramatically by manufacturer and product type. Most facilities require 1,000 to 3,000 units per batch for standard third-party manufacturing, with some going as low as 500 units for basic private label offerings.
Typical MOQ ranges:
| Manufacturing Model | MOQ Range (Units) | Best For |
|---|---|---|
| Private Label (basic) | 500 - 5,000 | Startups, market testing |
| Third-Party Manufacturing (standard) | 1,000 - 3,000 | Established small brands |
| Third-Party Manufacturing (custom) | 5,000 - 15,000 | Brands needing differentiation |
| Contract Manufacturing | 10,000 - 50,000 | Scale-oriented brands |

Fixed costs — machinery setup, raw material procurement, packaging, quality testing, regulatory compliance — must be distributed across enough units to be viable. The lower the batch size, the higher the per-unit cost.
Choose a manufacturer who can scale with demand before committing. Switching partners mid-growth disrupts production continuity, quality consistency, and customer experience.
Step 3: Research and Shortlist Manufacturers
Identify candidates through industry trade shows (Cosmoprof, in-cosmetics, MakeUp in), referrals, B2B platforms (Alibaba, ThomasNet), and region-specific directories.
Shortlist based on:
- Product category specialization — lipstick, emulsion, aerosol, and anhydrous products require different equipment
- Geography relative to target market — proximity affects lead times, logistics costs, and regulatory alignment
- R&D support vs. filling only — does the manufacturer help with formulation development or only execute your formula?
Check for client testimonials, production capacity, and any public regulatory violations or recall history. Cosmetics was the most frequently notified product category in the EU Safety Gate system in 2023, with 3,412 alerts — verify your shortlisted partners have clean compliance records.
Step 4: Verify Certifications and Regulatory Compliance
Key certifications a cosmetics manufacturer should hold:
- ISO 22716 (GMP for cosmetics) — baseline certification required globally
- FDA registration — required for products sold in the USA (under MoCRA)
- EU Cosmetics Regulation compliance — required for Europe (Responsible Person, CPSR, CPNP notification)
- BIS/CDSCO registration — required for India (Form COS-1 import registration)
Manufacturers compliant with EU GMP generally meet the most stringent global standards. Note that organic, halal, vegan, and dermatologically tested claims each require the facility — not just the formulation — to meet those standards.
Step 5: Protect Your Intellectual Property
IP protection must be addressed before sharing any formulation details.
Three layers of IP protection:
Sign a mutual NDA before disclosing any formulation specifics — the NDA should cover product formulations, ingredients, production processes, product designs, business plans, customer and supplier lists, and pricing data.
Include an explicit IP ownership clause in the manufacturing contract — the formulation, process, and all proprietary specifications remain the sole IP of the brand, including any tweaks made by the manufacturer during development.
Use process fragmentation or a platform intermediary — platforms like Distil manage multiple manufacturing partners while keeping full formulation data under the brand's ownership. No single partner has access to the complete formulation, structurally reducing IP leakage risk.

Contract clause for partnership termination: Define upfront whether the brand can take the formulation to another manufacturer if the partnership ends, and who owns any optimization made to the original brief.
In January 2026, NOMI Beauty sued Estée Lauder for trade secret misappropriation, alleging the cosmetics giant used proprietary systems to generate "billions" in revenue. NDAs and IP clauses are non-negotiable.
Step 6: Agree on a Benchmark Sample and Quality Standards
Before production begins, both parties must agree on a physical benchmark sample produced in the intended final packaging. This sample defines the acceptable range for texture, viscosity, color, fragrance intensity, and fill weight for every subsequent batch.
Why this matters: If a manufacturer changes their shea butter supplier and the product viscosity shifts, the benchmark sample is the contractual basis for raising a defect claim. Without a documented benchmark, quality disputes become subjective.
Store the benchmark sample in controlled conditions and reference it for every batch quality check.
Step 7: Establish Testing Protocols, Batch Codes, and Documentation
Types of testing to agree upfront:
- Stability testing — high-temperature, low-temperature, freeze-thaw cycling, photostability
- Microbial challenge testing — preservative efficacy
- Compatibility testing — packaging material interaction
- Safety assessments — CPSR for EU, safety substantiation under MoCRA for USA
Clarify which tests the manufacturer will run per batch and which are the brand's responsibility. ISO/TR 18811:2018 and Cosmetics Europe guidelines provide authoritative frameworks for cosmetic stability testing.
Batch code requirements:
Batch codes must appear on every finished unit — agree upfront on application method (printed, embossed, or labeled), placement on packaging, and how batch records will be retained and shared for traceability.
Under EU Regulation 1223/2009, Article 19(1)(e), a batch code is mandatory on the product label. Under U.S. regulations, 21 CFR 7.59(b) requires sufficient coding for positive product identification and effective removal of violative lots.
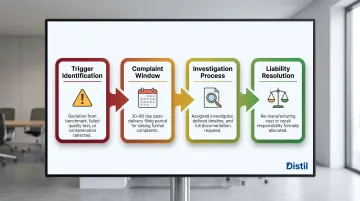
Step 8: Define Returns, Complaints Handling, and Escalation Procedures
The contract must clearly allocate financial liability for defective batches.
Define:
- What triggers a valid quality complaint (deviation from benchmark sample, failed stability test, microbial contamination)
- The window for raising a complaint (typically 30-90 days post-delivery)
- The process for investigation (who conducts it, timeline, documentation required)
- Who bears the cost of re-manufacturing or product recall
New brands frequently skip this clause — and it generates the most expensive disputes when something goes wrong. A contaminated batch requiring recall destroys both brand reputation and cash flow if liability is undefined.
For context, recall claims in topical products typically take more than 12 months to close, with business interruption coverage often continuing for 12-18 months post-incident.
How to Choose the Right Manufacturing Partner
Filter by Product Specialization
A manufacturer that specializes in color cosmetics uses entirely different equipment and expertise than one specializing in hair care emulsions. Working with a generalist to save cost often results in inferior texture, stability, or batch consistency.
Evaluate R&D Capability
Brands needing custom formulation should assess three things before committing:
- Whether the manufacturer has a dedicated in-house R&D team
- What their typical development and iteration timeline looks like
- Whether they have experience formulating for your target concerns and ingredient philosophy
Confirm Time-to-Market Alignment
Lead times vary dramatically by product type:
| Order Type | Total Turnaround | Key Phases |
|---|---|---|
| Private Label (standard stock) | 2-3 weeks (plus shipping) | Design approval, mold creation, production, QC |
| OEM / Custom Formulation | 3-5 months | R&D and prototyping, stability testing, packaging production, filling and assembly |
Confirm these timelines against your launch calendar before signing any agreement — a mismatch here is one of the most common reasons outsourcing relationships break down.
Full-Service vs. Pure Filler
Full-service manufacturing partners cover formulation development, raw material sourcing, production, QA, packaging, logistics, and compliance documentation under one engagement. This reduces the brand's coordination overhead and risk of miscommunication across multiple vendors.
Platforms like Distil represent this approach for personal care brands — operating an asset-light model that connects brands to a vetted network of manufacturing partners with a single point of contact. This model provides:
- Real-time visibility into batch yields, production status, and supply chain across all partner facilities
- Application-specific tuning and trials managed by a dedicated R&D team
- Customer-owned IP and data protected through structured access controls
Conduct a Facility Audit
Beyond certifications, request a facility audit or third-party audit report before committing to large volumes. A physical review of housekeeping standards, equipment calibration records, and batch documentation practices reveals operational discipline that a certificate alone cannot show.
Common Mistakes and Risks to Avoid
These three mistakes account for the majority of outsourcing failures — and each one is preventable with the right contractual and operational controls in place.
Sharing Formulation Details Without an NDA
Sharing formulation details before signing an NDA or IP protection agreement is the most costly mistake brands make. If the manufacturer replicates or shares the formula, you have no enforceable recourse. This risk is highest when brands rush partner selection to hit a launch deadline.
Choosing Based Solely on Lowest Unit Price
Cutting cost at the manufacturing stage often produces:
- Batch inconsistency across production runs
- Failed stability or challenge tests
- Ingredient substitutions made without brand notification
All of which cost far more to fix once a product is in market. Before committing, assess GMP compliance, ingredient sourcing standards, and quality control documentation — not just the unit price quote.

Failing to Define Ingredient Substitution Protocols
If a key ingredient becomes unavailable, an unconstrained manufacturer may substitute without approval — altering the product's texture, efficacy, or stability. The contract must require advance notification and written brand sign-off before any ingredient or supplier change is made.
Frequently Asked Questions
What is outsourcing in manufacturing?
Outsourcing in manufacturing means contracting an external specialist company to produce goods on behalf of a brand, using the brand's specifications. The brand retains IP and commercial control while the manufacturer provides production infrastructure and expertise.
Where is the best place to manufacture cosmetics?
The best location depends on your target market, required certifications, and cost structure. India offers competitive costs with growing GMP capability; Europe (Germany, Switzerland, Italy) is preferred for the highest regulatory compliance; China offers scale and speed. Each option carries different trade-offs in lead time, MOQ, and compliance complexity.
What is the difference between contract manufacturing and private label cosmetics?
Contract manufacturing produces goods to the brand's custom formulation and specifications — the brand owns the formula. Private label uses a pre-existing manufacturer formulation that multiple brands can purchase and rebrand. The key difference is IP ownership and the level of product customization.
How do I protect my formulation IP when outsourcing cosmetics manufacturing?
Protection works in three layers:
- Sign a mutual NDA before disclosing any formulation detail
- Include an explicit IP ownership clause in the manufacturing contract covering the formula and any modifications
- Use process fragmentation or a platform intermediary so no single manufacturer has access to the full formulation
What certifications should a cosmetics manufacturer have?
Key certifications to verify:
- ISO 22716 — GMP for cosmetics, the global baseline
- FDA registration — required for products sold in the US
- EU Cosmetics Regulation compliance — required for the European market
- Category-specific certifications (organic, halal, vegan, cruelty-free) — should match your product claims
How long does it typically take to launch a product through outsourced manufacturing?
Timelines depend on formulation complexity: using a pre-made private label formulation can reduce total lead time to 8-12 weeks; custom formulation development typically adds 3-6 months before production begins; total time from brief to first commercial batch for a fully custom product is commonly 6-12 months.


