
Introduction
For formulators and brand teams, ingredient selection is rarely straightforward. Balancing efficacy, safety, and stability is difficult at lab scale — and the complexity compounds at commercial volumes. Industry data puts quality control rejection rates at 15-25% of production volume due to formulation and scale-up issues, with active ingredient potency losses of 20-50% when processing isn't tightly controlled.
A skincare formulation is an engineered system. Chemistry, concentration, and compatibility all determine whether a product performs or fails — not just what's on the ingredient list.
This guide covers the essential building blocks of skincare formulation, core principles that separate effective products from ineffective ones, and the critical transition from lab to commercial production that determines whether your formula succeeds commercially.
TLDR
- Active ingredients — hyaluronic acid, niacinamide, retinol, vitamin C, peptides — must be selected based on skin concern, pH requirements, and stability profiles
- Emulsifier HLB values determine system type (oil-in-water vs. water-in-oil); base ingredient selection drives texture and stability
- Preservatives, surfactants, and solubilizers are mandatory for safety and shelf life in any water-containing formulation
- pH mismatches and poor storage can cost retinol 40–100% of its potency — concentration accuracy is critical at every stage
- Scale-up without validated equipment geometry and quality systems drives 15–25% rejection rates — dual-sourced ingredients reduce that risk
What Is Skincare Formulation?
Skincare formulation is the science of combining active, functional, and base ingredients into a stable, safe, and efficacious product. A formulation isn't a recipe — it's a designed system where every component has a purpose and interacts with others. Under EU Regulation EC 1223/2009 and the US Modernization of Cosmetics Regulation Act (MoCRA, 2022), cosmetic products are defined as preparations applied to skin, hair, or nails to cleanse, beautify, or alter appearance.
Every commercial formulation must achieve three properties:
- Efficacy — does it deliver the claimed benefit?
- Safety — does it avoid irritation, contamination, and instability?
- User experience — does it feel right and stay stable over its shelf life?
The global skincare ingredients market was valued at USD 13.4 billion in 2024 and is projected to reach USD 21.5 billion by 2034 — a growth rate that reflects how rapidly new ingredient categories and supply ecosystems are expanding to meet formulator demand.
That expansion also means more complexity. Moving from small-batch development to commercial production introduces sourcing variability, regulatory compliance requirements, and quality control challenges that are far harder to retrofit than to build in from the start.
The Building Blocks: Essential Skincare Ingredient Categories
Active Ingredients
Active ingredients are the performance core of any formulation — they target specific skin concerns and drive marketing claims. The most commercially significant actives include:
Niacinamide (Vitamin B3) is one of the few actives that combines genuine stability with broad efficacy. At 2-5% concentration, clinical trials show it increases ceramide synthesis by 4-5x and reduces melanosome transfer by 35-68% — addressing both hyperpigmentation and barrier function within a single ingredient. It remains stable to light and oxygen and tolerates most product categories, making it a low-risk, high-value addition to most formulations.
Hyaluronic Acid (HA) works differently depending on molecular weight. High-molecular-weight HA forms a hydrating film on the surface; low-molecular-weight fractions penetrate deeper into the epidermis. Multi-weight formulations that combine all three fractions show the strongest clinical results — surface hydration and deeper water-binding simultaneously.
Retinol presents a stability challenge that directly affects shelf-life claims. The EU restricts use to 0.3% in face products and 0.05% in body products, but concentration alone doesn't guarantee efficacy: retinoid content in commercial formulas has declined 0-80% after six months at 25°C and up to 100% at 40°C. Airless packaging, light-protective containers, and validated stability protocols aren't optional — they're prerequisite.
Vitamin C (L-Ascorbic Acid) is pH-dependent in a way that shapes the entire formula architecture. Below pH 3.5, it's stable and penetrates effectively. At pH 7, 99.9% exists in ionized form with negligible bioavailability. Derivatives like ascorbyl glucoside and sodium ascorbyl phosphate avoid this constraint but typically deliver lower efficacy than L-ascorbic acid.
Peptides degrade through heat, hydrolysis, and oxidation — so process order matters. They must be added during cool-down below 40°C. Formulators pairing peptides with retinol report meaningful improvements across wrinkles, elasticity, hydration, and barrier function compared to either ingredient alone.
| Active | Primary Mechanism | Effective Range | Key Stability Concern |
|---|---|---|---|
| Niacinamide | Ceramide synthesis, melanosome inhibition | 2-5% | Hydrolysis below pH 4.5 |
| Hyaluronic Acid | Hydration film, water binding | Molecular weight dependent | Generally stable |
| Retinol | Retinoic acid receptor activation | EU max 0.3% (face) | Photodegradation, thermal instability |
| Vitamin C (L-AA) | Collagen synthesis, antioxidant | pH-dependent penetration | Oxidation; requires pH below 3.5 |
| Peptides | Cell signaling for collagen/elastin | Sequence-specific | Heat degradation, hydrolysis |

Base and Structural Ingredients
Actives only perform as well as the base system that delivers them. Emulsifiers determine texture, skin feel, and which actives a formula can carry. Oil-in-water (O/W) emulsions use emulsifiers with HLB (Hydrophilic-Lipophilic Balance) values of 8-18, producing lighter textures that absorb quickly. Water-in-oil (W/O) emulsions use HLB values of 2-7, creating richer, more occlusive textures ideal for dry skin or barrier repair.
Common emulsifiers include glyceryl stearate citrate, cetearyl olivate/sorbitan olivate (Olivem 1000), and polyglyceryl esters for clean-beauty positioning.
Effective moisturization requires three complementary mechanisms working in sequence:
- Humectants (glycerin, hyaluronic acid, aloe vera) draw water from the environment and dermis into the stratum corneum
- Emollients (squalane, caprylic/capric triglyceride, cosmetic esters) fill gaps between corneocytes to smooth and soften skin texture
- Occlusives (petrolatum, dimethicone, beeswax, shea butter) form a physical barrier to reduce transepidermal water loss and seal in what the humectants and emollients deliver
Botanical Oils and Butters provide emolliency and nourishment. Argan oil contains 43-49% oleic acid and 29-37% linoleic acid, plus antioxidants and vitamin E. Jojoba oil is technically a liquid wax containing over 98% pure wax esters — it's noncomedogenic, hypoallergenic, and provides excellent lubricity without greasiness. Shea butter is rich in oleic and stearic acid, providing both emollient and occlusive properties.
Distil offers cosmetic esters including coco-caprylate/caprate (lightweight, silky feel), neopentyl glycol diheptanoate (smooth, non-greasy), and octyldodecanol (enhances spreadability) — all developed from plant-based inputs with application-specific sensory tuning.
Functional Additives: Preservatives, Surfactants, and Solubilizers
Preservatives are mandatory for any water-containing formulation to prevent microbial contamination. The choice depends on formulation pH, water activity, and brand positioning.
Common options and their constraints:
- Phenoxyethanol: broad-spectrum activity at 0.5-0.8%; maximum 1.0% under EU Cosmetics Regulation Annex V; typically combined with ethylhexylglycerin for enhanced efficacy
- Potassium sorbate: pH-dependent — functions optimally below pH 6.0; rarely used alone, usually paired with a complementary system
- Approved list: EU Annex V covers 60 approved preservatives with specified maximum concentrations and conditions of use
Preservation efficacy is validated through ISO 11930:2019 challenge testing: standardized microbial inocula are introduced and log-reduction is measured over 28 days.
Surfactants reduce surface tension to lift oil and dirt from skin, used in cleansers, shampoos, and body washes. Mild surfactant systems (coco-glucoside, sodium cocoyl isethionate) are preferred over sodium lauryl sulfate to minimize barrier disruption. Distil produces sucrose cocoate, a sugar-derived surfactant suited to sulfate-free scalp care shampoos, micellar waters, and luxury conditioning cleansers.
Solubilizers (polysorbate 20, PEG-40 hydrogenated castor oil) enable oil-soluble actives like essential oils or vitamin E to disperse uniformly in water-based formulas. Without solubilizers, oils separate or concentrate in pockets, creating irritation risk and product instability.
How to Use Skincare Ingredients: Core Formulation Principles
Phase Addition and Heat-Sensitive Actives
Standard emulsion formulation follows a three-phase process:
- Water phase: Water-soluble ingredients heated to 70-75°C
- Oil phase: Oil-soluble ingredients heated to matching temperature
- Cool-down phase: Heat-sensitive actives (retinol, vitamin C, peptides, fragrances, preservatives) added below 40°C
Incorporating heat-sensitive actives during heat processing degrades their efficacy and negates their value in the formula.
pH Management
Skin's natural pH sits between 4.5 and 5.5. Formulations outside this range risk disrupting the skin barrier, reducing active efficacy, or causing irritation. However, specific actives require divergent pH targets:
- Vitamin C is most stable and penetrates skin effectively at pH below 3.5
- AHAs are most effective below pH 4, where more acid exists in un-ionized (free acid) form
- Niacinamide is most stable at neutral pH (approximately 6.0) and may hydrolyze to nicotinic acid below pH 4.5
Regulatory thresholds vary by jurisdiction:
- US FDA: Recommends no more than 10% AHA at pH 3.5 or higher for consumer cosmetics
- EU SCCS: Recommends glycolic acid up to 4% at pH of at least 3.8 and lactic acid up to 2.5% at pH of at least 5.0
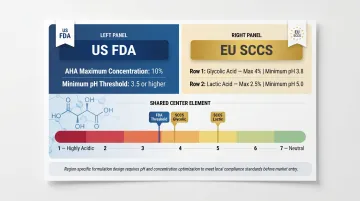
Product teams must design region-specific formulations to meet the stricter EU thresholds.
pH compatibility sets the foundation; the next layer is how ingredients interact with each other.
Ingredient Synergy and Antagonism
Certain combinations amplify benefits:
- Niacinamide + hyaluronic acid for barrier hydration — niacinamide supports ceramide synthesis while hyaluronic acid draws moisture into the stratum corneum
- Retinol + peptides for anti-aging — retinol drives cell turnover while peptides signal collagen production via different receptor pathways
Others interfere with each other:
- Ascorbic acid and niacinamide form a yellow-colored complex at elevated temperatures; at room temperature the reaction is slow, but formulators should still monitor color stability over shelf life
Formulators must test combinations — not just individual ingredients — before finalizing a formula.
Getting combinations right is only half the equation — the amounts used within those combinations matter just as much.
Concentration Accuracy
Using too little of an active produces no visible effect; too much increases sensitization risk and often violates regulatory limits. Most actives have clinically effective and/or regulatory-defined concentration ranges:
- Niacinamide: 2-5% for clinical benefits
- Retinol: EU max 0.3% (face), 0.05% (body)
- AHAs: US max 10%, EU max 4% (glycolic acid)
Formulators must work within these boundaries to ensure safety and efficacy.
Stability, Safety, and the 1% Rule
Stability Testing
Stability testing confirms a formula will perform as intended throughout its claimed shelf life. Three major categories exist:
Accelerated stability testing subjects products to 40°C ± 2°C at 75% RH for 6 months to simulate extended shelf life. Six weeks at these conditions can approximate 6 months of real-time aging.
Freeze-thaw cycling alternates between -10°C to -20°C and room temperature to stress-test emulsion integrity.
Real-time (long-term) testing holds products at 25°C ± 2°C for 12-24 months with periodic checks at 1, 3, 6, 9, 12, 18, and 24 months. ISO 29621 provides guidelines on stability testing of cosmetic products.
The 1% Rule
Under EC 1223/2009, Article 19, ingredients must be listed in descending order by weight. Ingredients in concentrations of less than 1% may be listed in any order after those in concentrations of more than 1%. This affects how formulas are disclosed, how competitors read product composition, and how brands strategically position key actives while protecting formulation IP.
Safety Testing Requirements
| Requirement | EU (EC 1223/2009) | US (MoCRA 2022) | India (Cosmetic Rules 2020) |
|---|---|---|---|
| Safety Substantiation | Mandatory; CPSR required | Mandatory; adequate safety substantiation | Required; safety data per BIS/IS |
| Facility Registration | Required with responsible person | Required; renewal every 2 years | Manufacturing/import license via state/CDSCO |
| Product Listing | CPNP | Annual listing with FDA including ingredients | Registration per Rule 12(1) before import |
| Adverse Event Reporting | Required | Serious events within 15 business days | Required |
| GMP | Recommended (ISO 22716) | FDA to establish mandatory GMP | Enforced by state licensing authorities |

Beyond registration, marketing claims carry their own substantiation requirements:
- Clinically tested: Requires testing on humans under qualified professional supervision
- Dermatologist tested: Requires a dermatologist to oversee the study design and execution
- Hypoallergenic: Requires demonstration of minimized allergenic potential, typically through Human Repeat Insult Patch Testing (HRIPT)
Quality Systems and Batch Consistency
A well-developed formula can still underperform commercially if ingredient quality shifts between batches or manufacturing partners operate without standardized controls. An R&D-integrated supply chain addresses this directly — unified quality systems applied across all production sites eliminate process drift before it reaches finished goods.
Distil's R&D team, with backgrounds spanning Dow, BASF, and L'Oréal, embeds these quality protocols across 20+ vetted manufacturing partners to keep batch-to-batch output consistent at commercial scale.
From Lab to Commercial Scale: What Skincare Brands Need to Know
Scale-Up Challenges
Scaling a formulation from lab batch to commercial production introduces new variables: mixing equipment type and shear force, heating and cooling rates, ingredient supplier variations, and filling line compatibility. A formula that works at 1 kg often requires reformulation or process adjustment at 100 kg or 500 kg.
Effective emulsification requires high shear rates of 5,000-15,000 s⁻¹ for initial droplet breakdown. Production equipment must be geometrically similar to lab-scale mixers; heat transfer characteristics change as batch size increases.
Common failure points at scale include:
- Phase separation from inadequate shear energy
- Texture/sensory defects from inconsistent droplet size (ranging from less than 1 micron to greater than 20 microns)
- Active ingredient degradation of 20-50% when processing is uncontrolled
- Air entrainment causing oxidation and cosmetic defects
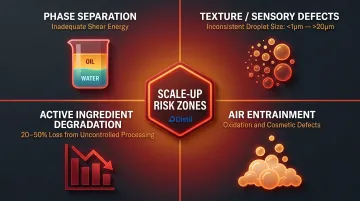
Brands must plan for scale-up trials as a distinct phase of development, not an afterthought.
Ingredient Sourcing Strategy
At commercial scale, brands need reliable, compliant, and consistent ingredient supply. Sourcing from multiple approved suppliers with verified COAs (certificates of analysis), MSDS documentation, and regulatory clearance is essential.
Supply chain vulnerabilities are real:
- Ukraine and Russia accounted for approximately 60% of global sunflower oil production; geopolitical conflict has strained availability
- The EU Deforestation Regulation is adding complexity to palm kernel oil and derivative sourcing (emulsifiers, surfactants)
- Climate change-driven droughts have caused consecutive years of low olive oil production
Dual-sourcing strategies and non-palm alternative qualification are now essential risk management decisions. RSPO-certified alternatives — such as coco-caprylate/caprate and sucrose cocoate — can replace palm derivatives across many formulations without compromising sensory performance.
End-to-End Integration
These sourcing and scale-up challenges are compounded when brands manage multiple vendors without visibility into batch consistency. An integrated approach — spanning formulation support, manufacturing execution, and supply chain oversight — reduces the handoff failures that drive 15-25% rejection rates during scale-up.
Distil's platform covers this end-to-end, with:
- Application-specific formulation support from a dedicated R&D team
- A network of 20+ vetted manufacturing partners across India
- Real-time production visibility and batch tracking
- A single point of contact from lab trial to commercial delivery
This structure lets brands scale without capital expenditure or IP exposure.
Frequently Asked Questions
What are the ingredients for skincare?
Skincare ingredients fall into three main categories: actives (hyaluronic acid, retinol, vitamin C, niacinamide, peptides), base ingredients (emulsifiers, botanical oils, butters, waxes), and functional additives (preservatives, surfactants, solubilizers). A serum targeting hyperpigmentation, for instance, will use a very different active and delivery system than a barrier-repair moisturizer.
What is the 1% rule in skincare?
The 1% rule is an INCI labeling convention under EC 1223/2009, Article 19. Ingredients used at 1% or below can be listed in any order after those present above 1%. This affects formulation transparency and IP protection considerations.
What are examples of personal care products?
Common personal care products include moisturizers, serums, cleansers, toners, sunscreens, body washes, shampoos, and lip balms. Each product type has a distinct formulation architecture and ingredient combination based on its intended function.
What is the difference between active and inactive ingredients in skincare?
Active ingredients deliver the primary skin benefit (for example, niacinamide for pore refinement or retinol for fine line reduction), while inactive or base ingredients support texture, stability, delivery, and shelf life without directly driving the product's core claim.
How do I ensure my skincare formulation is stable and safe?
Conduct stability testing (accelerated and real-time), perform preservative efficacy testing per ISO 11930:2019, and validate safety through irritation and patch testing before commercial launch. When selecting a manufacturing partner, verify that they apply consistent quality protocols across all production sites to avoid batch variability.


