
Introduction
Regulators across the EU, US, and India are tightening safety thresholds for petroleum-derived ingredients. Retailers are demanding clean-label certifications. Consumers are reading INCI lists and rejecting products with unfamiliar synthetic additives. For formulators in personal care, food, pharma, and industrial sectors, rethinking emulsifier choices is now a contractual requirement, not a future consideration.
Bio-based emulsifiers have moved from niche sustainability experiments to commercial necessities. As of February 2026, 71 EU food additives remain under re-evaluation, with several emulsifiers facing amended specifications or potential removal. The real value of switching shows up in compliance readiness, formulation performance, and supply chain resilience — well beyond environmental claims.
What follows is a practical breakdown for formulators and procurement teams: when bio-based emulsifiers make operational sense, where they perform, and what to evaluate before making the switch.
TL;DR
- Bio-based emulsifiers — derived from plant oils, sugars, and microbial fermentation — now match or outperform synthetic alternatives across most commercial applications
- Key advantages: lower regulatory risk, cross-sector formulation flexibility, and supply chain sustainability credentials that synthetic options can't match
- The global biosurfactants market hit USD 3.13 billion in 2023, with 6.1% CAGR projected through 2030
- The main challenge: grade selection and application-specific formulation — getting either wrong leads to stability failures and reformulation costs
What Are Bio-Based Emulsifiers?
Bio-based emulsifiers are substances derived from renewable biological feedstocks — vegetable oils, sugars, proteins, and microbial byproducts — that reduce interfacial tension between oil and water phases, enabling stable emulsions to form and persist.
ISO/DIS 21680 defines "bio-based" as "derived wholly or partly from biomass," establishing the standard for characterizing renewable content. Peer-reviewed literature divides these into two categories:
- First-generation: Chemically processed from plant feedstocks — alkyl polyglucosides (APGs), sucrose esters, and lecithin
- Second-generation: Biosynthesized by microorganisms through fermentation — sophorolipids and rhamnolipids
Common Bio-Based Emulsifiers
| Molecule | Source | Primary Sectors |
|---|---|---|
| Lecithin | Soy, sunflower, egg | Food, pharma, personal care |
| Alkyl polyglucosides (APGs) | Corn/coconut sugar + fatty alcohol | Personal care, household, industrial |
| Sucrose esters | Sucrose + fatty acids | Food, pharma, cosmetics |
| Mono- and diglycerides | Vegetable fats/oils | Food (bakery, dairy) |
| Gum arabic | Acacia tree exudate | Food, beverage |
| Sophorolipids | Yeast fermentation | Personal care, agriculture, bioremediation |
| Rhamnolipids | Bacterial fermentation | Industrial, enhanced oil recovery |

The same lecithin suitable for chocolate formulation differs substantially from the grade optimized for a pharmaceutical emulsion system — purity specification and concentration determine sector fit, not just the molecule itself.
For formulators, these molecules deliver on emulsion stability, texture, shelf life, biocompatibility, and regulatory clearance at the same time — performance that stands on its own, independent of sustainability positioning.
Key Advantages of Bio-Based Emulsifiers
The advantages discussed below are grounded in operational and commercial impact — outcomes that formulators, product managers, and procurement teams actively track: regulatory exposure, product quality, cost of compliance, and sourcing risk.
Advantage 1: Regulatory Compliance and Risk Reduction
Bio-based emulsifiers are substantially better positioned under current and emerging chemical regulations compared to petroleum-derived equivalents — including REACH (EU), TSCA (US), and India's BIS chemical safety frameworks — because they are biodegradable, lower in toxicity, and derived from non-fossil sources.
Brands using bio-based emulsifiers spend less time reformulating products in response to new restrictions, avoid costly ingredient substitutions under regulatory timelines, and can access markets (particularly in the EU) where specific synthetic emulsifiers face phase-out or labeling restrictions.
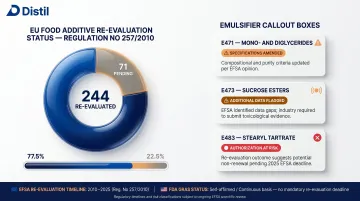
Under Regulation (EU) No 257/2010, EFSA is required to re-evaluate all 315 food additives authorized before January 2009. As of February 2026, 244 have been re-evaluated and 71 remain pending. Several emulsifiers have been directly affected:
- E471 (mono- and diglycerides): specifications amended
- E473 (sucrose esters): flagged with calls for additional data
- E483 (stearyl tartrate): no industry data submitted; authorization may be revised or removed
In personal care, the COSMOS standard prohibits all ingredients derived from petrochemistry and produced via ethoxylation, affecting 600+ substances used in conventional cosmetics. Bio-based emulsifiers (APGs, sucrose esters, lecithin) are compliant alternatives.
Proactive regulatory alignment reduces reformulation costs and protects time-to-market. Contrast this with brands that wait for restrictions and are forced into reactive, expensive reformulations. FDA food additive petitions require 1-3 years; GRAS notifications take 9-14 months. Regulatory non-compliance can delay launches, trigger retailer de-listing, and expose companies to reputational damage.
KPIs impacted:
- Compliance cost per SKU
- Regulatory approval timelines
- Number of restricted ingredient substitutions required per year
- Clean-label certification eligibility
This advantage is most critical for brands with EU, US, or APAC distribution where chemical regulations are tightening; for product lines carrying "clean label," "natural," or "COSMOS-certified" claims; and for manufacturers responding to retailer ESG procurement mandates.
Advantage 2: Formulation Versatility and Performance Across Applications
Bio-based emulsifiers — including lecithin, sophorolipids, gum arabic, mono- and diglycerides, and modified starches — deliver stable emulsification across a broad range of pH levels, temperatures, and formulation types (oil-in-water and water-in-oil), when matched to application pH and HLB requirements.
Bio-based emulsifiers interact with proteins (improving dough elasticity in bakery applications), stabilize fat droplets in personal care emulsions, reduce surface tension in pharmaceutical drug delivery systems, and form protective interfacial layers in industrial emulsions. The common thread is their amphiphilic structure, which can be tuned — through esterification degree, chain length, and HLB — to match the demands of each application.
Performance benchmarks:
Replacing 25% of SLES with decyl glucoside (APG) reduced skin irritation by 20-30%, while APGs as co-surfactants enabled a 20% reduction in total surfactant concentration without loss of foaming performance. Sucrose ester CMC values are 1-2 orders of magnitude lower than commercial synthetic surfactants, indicating higher efficiency at lower concentrations.
Sophorolipids maintain stability across pH 3-11 and high salinity conditions, with cell viability above 85% in cytotoxicity testing — indicating low irritation potential.
HLB coverage:
| Emulsifier | HLB Range | Emulsion Type |
|---|---|---|
| Sorbitan esters | 1-8 | W/O |
| Sucrose esters (di/triesters) | 5-7 | W/O |
| Lecithin | ~7 | W/O to O/W |
| Sucrose esters (monoesters) | >11 | O/W |
| Alkyl polyglucosides (APGs) | >10 | O/W |
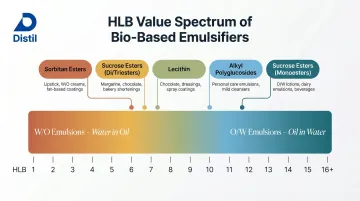
Bio-based emulsifiers collectively cover the full HLB spectrum (approximately 1-16+), enabling both W/O and O/W formulations without petrochemical ingredients.
Multi-sector applicability gives formulators flexibility to standardize sourcing across product lines rather than maintaining separate synthetic emulsifier inventories. Consistent emulsification performance — HLB flexibility, low migration, thermal stability — directly reduces batch rejections and reformulation iterations.
KPIs impacted:
- Emulsion stability index
- Batch rejection rates
- Number of reformulation iterations
- HLB flexibility range
- Concentration efficiency (active dosage needed per batch)
Highest impact in multi-product or multi-sector formulation environments; in sensitive end-use categories (food-contact, skin-contact, drug delivery) where both performance and safety profile must meet stringent requirements simultaneously.
Advantage 3: Supply Chain Sustainability and Resilience
Bio-based emulsifiers are derived from renewable feedstocks — plant oils (soy, sunflower, coconut, palm), sugars, and microbial biosynthesis — meaning their production is less exposed to the petroleum price volatility and crude supply disruptions that historically drive synthetic emulsifier cost swings.
Three operational benefits follow from this:
- Diversified feedstock sourcing reduces single-supplier dependency
- Biodegradable end-products simplify waste handling and effluent compliance
- Lower carbon footprint across the production lifecycle supports Scope 3 ESG reporting
APGs achieved 88% degradation in the Closed Bottle Test (OECD pass level: 60%) and 90% in the DOC Die-Away Test (pass level: 70%). Sucrose laurate degraded within 1 day — these metrics satisfy Scope 3 and ESG reporting requirements.
Market growth signals:
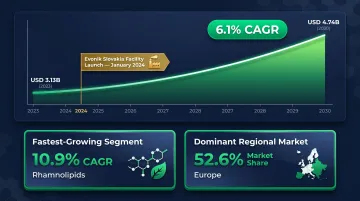
The biosurfactant market demonstrates strong commercial momentum:
- Global market at USD 3.13 billion in 2023, projected to reach USD 4.74 billion by 2030 at 6.1% CAGR
- Rhamnolipids are the fastest-growing segment at 10.9% CAGR
- Europe holds 52.6% of the global market
Multiple suppliers now offer industrial-scale quantities. In January 2024, Evonik commissioned the world's first industrial-scale rhamnolipid biosurfactant facility in Slovakia, producing 100% biobased and 100% biodegradable product from European corn sugar.
Supply chain sustainability translates into cost predictability — biological feedstock costs are generally less correlated to crude oil price swings than petrochemical-derived emulsifiers — and into commercial differentiation. Brands that demonstrate sustainable ingredient sourcing gain access to ESG-conscious retail channels and institutional buyers. Unilever has committed to making all product ingredients achieve "ultimate biodegradation" by 2030, driving reformulation across their surfactant and emulsifier systems.
KPIs impacted:
- Carbon footprint per kg of emulsifier (Scope 3 emissions)
- Supply chain concentration risk score
- ESG/sustainability audit ratings
- Renewable content percentage
- Biodegradability rating
Most impactful for brands with net-zero commitments, ESG-rated procurement processes, or consumer-facing sustainability claims; also highly relevant during periods of petrochemical price volatility or geopolitical disruption to fossil fuel supply chains.
What Happens When Brands Rely on Synthetic Emulsifiers Too Long
Delayed adoption carries real costs. Brands face regulatory catch-up expenses from forced reformulation under time pressure, restricted market access as EU chemical safety rules tighten, and eroding clean-label positioning as competitors transition ahead of them.
Operational risks specific to synthetic emulsifier dependence:
- Exposure to ingredient phase-out without lead time for reformulation
- Higher long-term compliance monitoring costs as regulatory scrutiny intensifies
- ESG scoring penalties that affect institutional investor ratings and retailer sustainability assessments
- Difficulty scaling into markets where bio-based content thresholds are becoming a procurement prerequisite
E483 (stearyl tartrate) received no industry data submissions during EFSA's re-evaluation call, triggering revision of its authorization and potential removal from the EU approved list. Formulators relying on this additive face forced reformulation if authorization is withdrawn.
The commercial exposure here is as real as the regulatory one. Slow transitions create reformulation backlogs, delayed product launches, and widening gaps between what brands claim on-pack and what their formulations actually contain — a position that becomes harder to recover from as the market moves on.
How to Get the Most Value from Bio-Based Emulsifiers
Bio-based emulsifiers work best when selection is application-specific: the grade, source, concentration, and processing conditions must be matched to the emulsion type, end-use requirements, and regulatory jurisdiction. A lecithin suitable for chocolate formulation is not the same as a sophorolipid optimized for a personal care emulsion system.
Three critical success factors:
- Consistent batch quality from a qualified supplier — batch-to-batch variation can undermine formulation stability
- Application-specific formulation trials — validate stability under real processing conditions, not just theoretical compatibility
- Ability to scale from lab to production without performance drift — this is where many transitions fail

All three hinge on supplier capability. Distil addresses each directly:
- R&D-led trials: A dedicated team with backgrounds at Dow, BASF, L'Oréal, Huntsman, and Reliance conducts application-specific formulation and validation
- Scalable manufacturing: Production runs through 20+ partner facilities with a unified quality system that prevents process drift across batches
- Single point of contact: One team manages the journey from lab to commercial scale, with real-time visibility into batch yields
For example, Distil's sucrose cocoate — a plant-based mild surfactant manufactured from RSPO-certified feedstocks — uses specialized transesterification processes to prevent sucrose caramelization, preserving a high natural origin index. That technical precision matters when positioning a product for clean-label personal care markets.
Switching to bio-based isn't a one-time project — it's an ongoing process. Once you've validated a formulation, build in regular checkpoints:
- Review formulation performance against benchmarks as feedstock sources evolve
- Update supply chain documentation to reflect bio-based content for ESG reporting
- Track regulatory developments to stay ahead of the next wave of ingredient restrictions
Conclusion
Bio-based emulsifiers have moved well past proof-of-concept. They offer measurable advantages in regulatory positioning, formulation performance, and supply chain resilience — advantages that grow more valuable as ingredient scrutiny increases across global markets.
What determines whether that value is captured is application-specific execution: selecting the right grade, formulating with technical support, and scaling without compromising consistency. Distil's R&D-led approach — from lab trials to commercial production — is built to support exactly that transition, reliably and at scale.
Frequently Asked Questions
What is the difference between bio-based emulsifiers and biosurfactants?
Both are derived from biological sources and reduce surface tension, but biosurfactants are produced entirely by microbial processes with no traditional chemical reactions, while bio-based emulsifiers can include plant-derived or chemically modified biological molecules. Bio-based emulsifiers typically have higher molecular weights and are primarily used to stabilize emulsions, while biosurfactants focus on surface tension reduction.
Are bio-based emulsifiers as effective as synthetic emulsifiers?
When matched correctly to the application — right grade, concentration, and processing conditions — bio-based emulsifiers perform comparably to synthetic alternatives, and in some cases outperform them on biocompatibility, low-concentration efficacy, and pH/temperature stability. Performance depends heavily on correct formulation and application-specific trials.
Which industries use bio-based emulsifiers most?
The largest markets are food and beverage, personal care and cosmetics, pharmaceuticals, and industrial applications such as agricultural formulations and oil processing. Personal care and food lead by volume, with personal care growing at 6.3% CAGR.
What are the most common natural sources of bio-based emulsifiers?
Key feedstocks include soy and sunflower lecithin, coconut oil derivatives, sugar-based molecules (sophorolipids, alkyl polyglucosides), plant gums (gum arabic), and microbial fermentation products. Feedstock choice affects the HLB value, performance range, regulatory status, and environmental footprint of the final emulsifier.
Do bio-based emulsifiers cost more than synthetic alternatives?
Some bio-based emulsifiers carry a cost premium, particularly those derived from fermentation or specialized plant sources. However, this cost must be weighed against long-term savings from reduced regulatory compliance costs, and lower long-term reformulation risk as regulations tighten. The cost gap is narrowing as production scales and manufacturing capacity expands.
Are bio-based emulsifiers safe for use in food and personal care products?
Many bio-based emulsifiers carry GRAS status (FDA) and are approved under EU food additive regulations — lecithin (21 CFR 184.1400), mono- and diglycerides (21 CFR 184.1505), and gum arabic (21 CFR 184.1330) are all affirmed GRAS with no usage limits beyond GMP. Each ingredient and application should still be verified against the relevant regulatory framework before use.


