
Introduction
For procurement leads, R&D heads, and product managers in personal care, advanced polymers, flavours and fragrances, and FMCG packaging, custom synthesis is a regular commercial decision — not an academic one. Choosing to build a compound to exact specifications, rather than sourcing from a standard catalogue, has direct consequences for supply chain resilience, product differentiation, and IP protection.
CRO (Contract Research Organization) services provide the external scientific and operational infrastructure that makes custom synthesis viable. They handle route design, feasibility assessment, small-scale synthesis, analytical validation, and — in integrated models — the transition to commercial production.
Both terms appear regularly in vendor conversations, but the operational reality behind them is rarely spelled out. This article explains what custom synthesis and CRO services actually involve, where they add genuine value, and where they don't.
TL;DR
- Custom synthesis means manufacturing a specific compound or formulation to order — defined purity, scale, and specification
- A CRO handles research and synthesis; a CMO handles large-scale manufacturing; many projects require both, sometimes through a single integrated partner
- Companies outsource to access specialist R&D, scale without capex, protect IP, and shorten the path from concept to commercial batch
- The process moves through route design, small-scale synthesis, analytical validation, and scale-up — with active collaboration required at every stage
- Success depends on brief quality, scale trajectory alignment, IP protection structures, and whether the CRO's expertise matches the chemistry type
What Is Custom Synthesis — and Where Do CRO Services Fit?
Custom synthesis is the design and production of a chemical compound or formulation that does not exist in a supplier's standard catalogue. It is manufactured exclusively for a specific client, to their stated structure, purity, quantity, and application requirements.
The outcome is a reproducible, application-ready compound or formulation, produced to a validated method, that a client can use directly in their product, process, or research pipeline.
Custom synthesis differs from two common alternatives:
- Catalogue procurement: Off-the-shelf supply of standardised compounds at fixed specifications
- Toll manufacturing: Production using the client's own recipe on a third-party asset — the client provides the formula, the toll manufacturer provides equipment and labour
In custom synthesis, the CRO co-develops or independently develops the synthesis route and method. The client specifies what they need; the CRO determines how to make it.
Building this capability in-house is expensive. It requires specialist talent — organic chemists, polymer scientists, formulation scientists — along with dedicated lab infrastructure, analytical equipment, and quality systems. For most brands operating in personal care, polymers, F&F, or FMCG, that is a non-core investment. Outsourcing transfers it to a focused specialist.
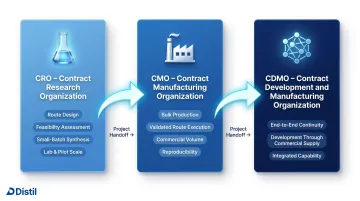
CRO, CMO, and CDMO — What the Terms Actually Mean
A CRO (Contract Research Organization) provides research and synthesis services at lab and pilot scale. Their core competency is solving the "how to make" problem — route design, feasibility assessment, small-batch synthesis. They are not optimised for high-volume production.
A CMO (Contract Manufacturing Organization) handles bulk production of an established compound at scale, executing a validated synthesis route reproducibly at commercial volume. A CDMO (Contract Development and Manufacturing Organization) combines both, offering continuity from development to commercial supply.
In specialty chemicals outside pharma — personal care, F&F, advanced polymers — these terms are used loosely. What matters operationally is whether the partner can handle your chemistry type, your required scale trajectory (from grams to commercial batch), and your regulatory or quality documentation requirements. The label matters less than the capability.
Why Specialty Chemical Brands Outsource Custom Synthesis to CROs
Economic Rationale: Fixed Cost to Variable Spend
Building internal synthesis capability requires lab infrastructure, specialist headcount (PhDs, MScs in organic/polymer/formulation chemistry), equipment, and quality systems. For most brands operating in personal care, polymers, F&F, or FMCG, this is a non-core investment.
Capital expenditure for chemical manufacturing equipment represents a multi-million dollar commitment. One modeled chemical manufacturing startup shows total capex of approximately $4.08 million for core equipment alone — including a $1.5M production reactor and an $800K distillation unit.
The financial picture for in-house chemical R&D is tight across the board:
- Chemical companies allocate 2-3% of annual sales toward R&D on average, with some reaching 8-9%
- Total US chemistry R&D investment stands at $14.8 billion
- Capital expenditure fell 8.4% year-over-year in 2024, with net profit margins averaging just 5.8% between 2000 and 2020
Outsourcing converts fixed R&D cost into a variable, project-by-project spend — no headcount, no equipment depreciation, no idle capacity.
Depth of Synthesis Knowledge That Takes Years to Build
CROs working across multiple clients and compound classes accumulate synthesis knowledge, analytical methods, and process optimisations that an internal team working in a narrower domain rarely develops. This is especially relevant for complex chemistries — unusual polymer architectures, personal care actives, and flavour compounds with tight sensory specifications.
Distil's R&D team includes PhDs, MScs, and engineers with 45+ years of combined experience from Dow, BASF, L'Oréal, Huntsman, and Reliance Industries. That depth translates directly into faster route design and fewer failed iterations — without the client having to build or staff an equivalent function internally.
Scalability Without Capital Expenditure
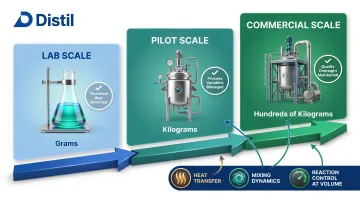
A CRO with a broad manufacturing partner network can move a client from a 50-gram proof-of-concept batch to a 500-kilogram commercial run without requiring the client to invest in plant or machinery.
Indian CDMOs can reduce manufacturing costs by 30% to 70% compared to MNCs manufacturing in-house. In agrochemicals, major MNCs currently outsource more than 70% of their production, according to industry analysis by Sathguru.
The asset-light model enables rapid scale-up while maintaining quality and consistency — without the client financing or operating production facilities. That same flexibility is what makes IP protection structurally viable: the client never has to hand the full picture to any single external party.
IP Protection Built Into the Operating Model
The client retains ownership of the synthesis outcome and the product data. A well-structured CRO relationship uses NDAs, process fragmentation (no single manufacturing partner sees the full formulation), and permission-controlled access to prevent IP leakage.
This is particularly important in competitive categories like personal care actives or proprietary polymer blends.
Distil structures its manufacturing model on exactly this principle: a control layer and fragmented partner access ensure that customer IP is never exposed to a single external party. Clients retain full ownership of their data, with security maintained through process segmentation and permission-based access.
Extended Workbench: Concurrent R&D Programs
Outsourcing allows brands to run concurrent R&D programs without capacity constraints. An external CRO can be activated for a project and wound down without staff restructuring — a real advantage when launching multiple product lines simultaneously or validating chemistry in a new segment before committing internal resources.
How Custom Synthesis and CRO Services Work, Step by Step
A custom synthesis engagement follows a structured progression: technical brief, route design, small-scale synthesis, analytical validation, iterative refinement, and scale-up. The full timeline runs anywhere from a few weeks to several months, depending on chemistry complexity and target batch size.
What the CRO needs from the client at the outset:
- Target compound structure or formulation parameters
- Intended application and performance requirements
- Purity specification
- Required quantity and likely scale trajectory
- Timeline
- Regulatory or documentation constraints (e.g., REACH compliance, COSMOS certification for personal care, food-grade standards for F&F)
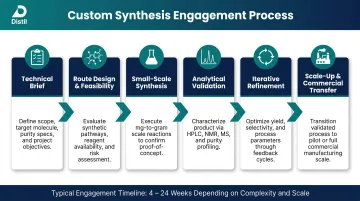
Step 1: Route Design and Feasibility Assessment
Before any chemistry begins, the CRO evaluates available synthesis pathways for the target compound — assessing yield potential, reagent availability, environmental and safety risk, cost profile, and scalability.
This stage may result in a go/no-go recommendation.
Two approaches exist:
- Reproducing a known synthetic route from literature or patents — faster, lower risk
- Designing a new route from scratch — longer, but produces a proprietary compound or formulation
Route scouting "identifies potential synthetic challenges early, allows for contingency planning, and helps select the most robust process for scale-up," according to BOC Sciences.
Step 2: Laboratory-Scale Synthesis and Analytical Validation
Synthesis begins at gram or tens-of-grams scale in a controlled lab environment, with iterative refinement of conditions:
- Temperature
- Solvent systems
- Catalyst selection
- Reaction time
The goal: a reproducible, high-purity output.
Every batch is characterized using analytical techniques matched to the compound class:
| Technique | Primary Use |
|---|---|
| NMR (Nuclear Magnetic Resonance) | Structural confirmation, isomer differentiation |
| HPLC (High-Performance Liquid Chromatography) | Purity determination, impurity profiling |
| GC (Gas Chromatography) | Volatile compound analysis, residual solvent testing |
| MS (Mass Spectrometry) | Molecular weight confirmation, structural elucidation |
Results are documented in a batch record and certificate of analysis. This is the stage where most technical risk is resolved before any capital is committed to scale-up.
Step 3: Scale-Up and Commercial Transfer
Once lab protocols are validated, they move to pilot-scale and then commercial-scale production.
This step introduces new variables:
- Heat transfer
- Mixing dynamics
- Reaction control at volume
In an asset-light CRO model, this step involves transferring the validated process to a manufacturing partner with appropriate equipment. The CRO maintains quality oversight and consistency controls across the transition.
Key Factors That Shape Custom Synthesis Outcomes
Quality of the Technical Brief
A vague or incomplete technical brief forces iteration cycles that add cost and delay. The more precisely a client defines the compound and its requirements upfront, the more efficiently the CRO can design the right synthesis route.
Clients should specify:
- What the compound is
- What it needs to do
- In what application
- Under what performance conditions
Chemistry Complexity Affects Timelines and Cost
Multi-step syntheses, chiral compounds, moisture or oxygen-sensitive reactions, and novel polymers require more specialized equipment and expertise than simpler transformations.
A Science perspective article confirms that "renewable feedstocks and fewer process steps can reduce financial and environmental costs" in chemical manufacturing. The search for structural complexity has led to manufacturing routes of increasing step count, each step adding cost and waste.
Cumulative yield impact: In a multi-step synthesis, overall yield equals the product of individual step yields. A 10-step synthesis with 90% yield per step produces only approximately 34% final yield. At 70% yield per step, the final yield drops to approximately 2.8%. This geometric compounding is the primary mechanism driving COGS in specialty chemical manufacturing.
Scale Trajectory and Partner Selection
Synthesis complexity doesn't just affect early-stage costs — it shapes which partners can realistically take a compound to commercial scale.
A CRO optimized for milligram-to-gram research samples is not the right partner if the client anticipates multi-hundred-kilogram commercial demand within 18 months. Route design, solvent choices, and process economics need to reflect the end scale from the start.
When a CRO has an integrated network of manufacturing partners, this transition is managed without the client re-briefing a different supplier from scratch.
Common Misconceptions About Custom Synthesis and CRO Services
Misconception: CRO Services Are Only for Pharma
Custom synthesis is widely used across:
- Personal care: Novel actives, emollients, preservatives
- Advanced polymer development: Masterbatches, functional additives
- Flavours and fragrances: Aroma molecules, essential oils
- Agrochemical formulations: Active ingredients, intermediates
- Specialty industrial chemicals: Application-specific compounds
For example, Amyris signed a multiyear agreement with International Flavors & Fragrances (IFF) to develop fragrance ingredients made by microbes from sugar. Givaudan and Firmenich also have fragrance deals with Amyris.
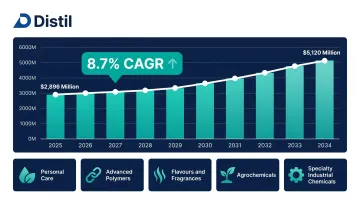
The custom synthesis service market was valued at $2,896 million in 2025 and is projected to reach $5,120 million by 2034 at a CAGR of 8.7%, driven by demand from agrochemicals, materials science, and biotechnology alongside pharma.
If application-specific chemistry is required and internal synthesis isn't a core capability, custom synthesis is worth evaluating — regardless of the industry.
Misconception: Outsourcing Synthesis Means Losing Control or IP
A properly structured CRO engagement includes:
- Perpetual NDAs — not the standard 3–5 year confidentiality clauses that can leave trade secrets exposed
- Explicit data ownership clauses so the client retains all experimental and process data
- Process fragmentation so that no single external party holds the complete formulation
According to an ACS webinar featuring partners from Goodwin Procter LLP, trade secrets require perpetual confidentiality obligations. If the secrecy obligation expires, trade secret status may be legally lost.
The client retains full data ownership and can transfer the validated process to another manufacturer if required.
Misconception: CRO Services Are Appropriate for Every Synthesis Need
CRO services add the most value when the chemistry is non-standard, the route is not yet defined, or the client needs to move from idea to validated compound without internal lab resources. That said, not every synthesis project is the right fit. CRO services may not be the right tool when:
- The compound is already commercially available at acceptable purity and price from a catalog supplier
- Internal chemistry capability already exists and has spare capacity
- The volume required is so large from the outset that a direct CMO relationship is more efficient
- The synthesis is straightforward enough that a toll manufacturer with the client's own recipe is the right model
Choosing the right model — CRO, CMO, toll manufacturer, or catalog supplier — depends on where the chemistry actually sits relative to your internal capabilities and commercial timeline.
Frequently Asked Questions
What is custom synthesis manufacturing?
Custom synthesis manufacturing is the process of producing a specific chemical compound or formulation to order, built to a client's defined structure, purity, scale, and application requirements. It is distinct from purchasing a standard off-the-shelf chemical product.
What is synthesis in manufacturing?
Synthesis in manufacturing refers to building a target chemical compound from simpler starting materials through controlled chemical reactions. In a manufacturing context, this means doing so reproducibly, at defined quality, and at a commercially viable scale rather than in a research setting.
What is CMC in pharma manufacturing?
CMC stands for Chemistry, Manufacturing, and Controls — a regulatory documentation framework in pharmaceutical development that covers how a drug substance is made, its quality specifications, and how consistency is controlled. CMC is pharma-specific and distinct from the broader concept of custom synthesis, which applies across specialty chemicals sectors well beyond pharma.
What is the difference between a CRO and a CMO in specialty chemicals?
A CRO (Contract Research Organization) handles compound design, route development, and small-to-pilot-scale synthesis: solving the "how to make it" problem. A CMO (Contract Manufacturing Organization) takes over for large-scale, ongoing production of an already-validated compound — most specialty chemicals projects engage a CRO first, then transfer to a CMO for commercial supply.
How does IP protection work in custom synthesis services?
IP protection in custom synthesis operates through NDAs with perpetual confidentiality obligations and client data ownership clauses. In more sophisticated models, process fragmentation ensures no single manufacturing partner sees the complete formulation or synthesis route, preventing independent replication by any external party.


