
Introduction
Most thermoplastics in their base form are brittle, flammable, UV-sensitive, or mechanically limited. Raw polyethylene degrades under sunlight within months. Neat polypropylene cracks under impact in cold conditions, and unmodified PVC ignites easily and emits toxic smoke when burned. These gaps between raw polymer and application-ready material are closed through polymer additives and fillers — the formulation tools that make engineered plastics possible.
Additives and fillers serve different but complementary roles:
- Stabilizers protect against thermal degradation during melt processing
- Flame retardants enable plastics to meet fire safety codes in automotive and electronics
- Fillers like glass fiber increase tensile strength by more than 200%
- UV absorbers extend service life of agricultural films and outdoor components
Together, they bridge the gap between a commodity resin and a material fit for demanding applications.
This guide covers the primary categories of polymer additives and fillers, how they work, how to select them, and where they are most critical across industrial applications.
TLDR
- Additives (stabilizers, flame retardants, plasticizers) modify polymer behavior at low loadings, typically under 5 wt%
- Fillers (calcium carbonate, glass fibers, talc) are inorganic particles or fibers added at higher volumes to reinforce structure or reduce cost
- Filler shape determines property improvements: fibers boost tensile strength, plates improve barrier, spheres reduce warp
- Selection depends on polymer base, processing method, end-use environment, and regulatory requirements
- Coupling agents and dispersion quality are critical; poor filler-polymer bonding degrades rather than improves properties
Additives vs. Fillers: What's the Difference?
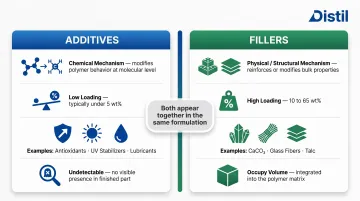
Polymer additives are chemical compounds introduced at low concentrations — typically under 5 wt% — to change how a polymer behaves during processing or in its end-use environment. They do not bulk up the material but alter its chemistry or surface behavior. Examples include antioxidants that prevent oxidative chain scission, UV stabilizers that absorb harmful radiation, and lubricants that reduce melt viscosity during extrusion. At these concentrations, they're often undetectable in the finished part.
Fillers are typically inorganic particles or fibers added in larger volumes — often 10–65 wt% — to physically modify the polymer matrix. They can reduce formulation cost through volume extension, add bulk, or significantly improve mechanical or thermal properties. Examples include calcium carbonate (CaCO₃) for stiffness and opacity, glass fibers for structural reinforcement, and talc for heat deflection. Fillers occupy space in the polymer matrix and contribute to the material's physical structure.
The key distinction: additives work through chemical mechanisms at low loadings; fillers are structural components added in much higher volumes. In practice, both appear together in the same formulation. A talc-filled polypropylene automotive dashboard compound might also contain thermal stabilizers, UV absorbers, and lubricants — each addressing a different performance requirement.
Major Types of Polymer Additives
Stabilizers (Thermal, UV, and Antioxidants)
Polymer chains degrade when exposed to heat, oxygen, or light. Thermal stabilizers prevent chain scission during melt processing, where temperatures often exceed 200°C. Antioxidants inhibit autoxidation — a chain reaction that weakens the matrix over time, particularly in polyolefins. UV stabilizers protect outdoor or light-exposed applications.
UV stabilizers fall into two categories:
- UV absorbers — absorb UV radiation and dissipate it as heat
- HALS (Hindered Amine Light Stabilizers) — function via a regenerative cycle involving the piperidyl ring structure, scavenging free radicals formed during photodegradation
HALS are particularly effective in polyolefins. The global UV light stabilizers market is projected to reach USD 2.81 billion by 2034, driven by automotive exterior and packaging applications where outdoor exposure is constant.
Without them, a polypropylene bumper would fade and crack within a year.
Flame Retardants
Most thermoplastics ignite readily. Flame retardant additives delay ignition, reduce smoke generation, and slow flame spread. The market is served by three primary chemistries:
- Halogenated (bromine/chlorine-based) — interrupt the combustion cycle by releasing halogen radicals that quench free radicals in the flame
- Phosphorus-based — promote char formation, creating a protective barrier
- Mineral-based (ATH, MDH) — decompose endothermically, releasing water vapor that dilutes combustible gases and cools the polymer
ATH (aluminum trihydrate) decomposes at approximately 200°C. MDH (magnesium hydroxide) decomposes above 300°C, making it suitable for polymers processed at higher temperatures. Both require high loading levels — typically 40-65 wt% — to achieve flame retardancy, which typically reduces impact strength and tensile modulus.

The global flame retardant chemicals market is projected to grow from USD 5.9 billion in 2024 to USD 8.0 billion by 2030 at a CAGR of 5.3%. Regulatory pressure from EU RoHS and REACH is accelerating reformulation away from halogenated to halogen-free systems in wire & cable and electronics.
Plasticizers
Where flame retardants harden and protect, plasticizers do the opposite: they soften. Typically non-volatile liquid organics — commonly phthalate or non-phthalate esters — plasticizers insert between polymer chains to increase chain mobility, reducing hardness and improving flexibility. Most commonly used in PVC, they transform rigid pipe-grade resin into flexible wire casing.
Loading level directly controls flexibility:
| Application | Loading (phr) | Shore A Hardness |
|---|---|---|
| Semi-rigid | 25-40 | 90-95 |
| Standard flexible | 45-65 | 70-85 |
| Highly flexible | 70-100+ | 50-70 |
Note that below 15 phr, "antiplasticization" can occur — counter-intuitively increasing stiffness rather than reducing it.
Regulatory restrictions are reshaping the market. DEHP, BBP, DBP, DIBP, and DCHP are listed as Substances of Very High Concern under EU REACH. US CPSIA bans eight phthalates at >0.1% in children's products. Medical and packaging compounders are reformulating to DINCH, ATBC, DEHT, or TOTM alternatives — DINCH in particular shows eightfold lower migration than DEHP in medical applications.
Lubricants and Anti-Blocking Agents
Internal lubricants reduce intermolecular friction within the melt to improve flow. External lubricants reduce adhesion between the melt and processing equipment surfaces, preventing sticking to screws and dies.
Anti-blocking agents (e.g., silica, diatomaceous earth) are added to film-grade polymers to prevent surface adhesion between layers during winding and storage. Critical in flexible packaging where film layers stack for thousands of meters on a roll.
Antistatic Agents and Colorants
Antistatic agents dissipate or prevent static charge buildup, important in electronics packaging and clean-room environments where electrostatic discharge can damage components or attract dust.
Colorants include:
- Pigments — insoluble particles that scatter light
- Dyes — soluble organics that absorb specific wavelengths
Colorant compatibility with the base resin and processing temperature must be validated. Compounders typically prefer pre-compounded masterbatches for consistent color distribution and easier dosing.
Types of Polymer Fillers and Their Effects
Calcium Carbonate (CaCO₃)
The most widely used filler globally, derived from limestone (ground calcium carbonate, GCC) or synthesized (precipitated calcium carbonate, PCC). Primary roles include:
- Cost reduction through volume extension
- Improved stiffness
- Enhanced whiteness and opacity (reducing need for white masterbatch)
- Improved surface gloss in film applications
Widely used in PVC, polyolefins, and unsaturated polyesters. The global calcium carbonate market is projected to grow from USD 40.72 billion in 2026 to USD 56.24 billion by 2034.
Surface coating matters. Stearic acid coating reduces surface tension from approximately 210 to 40-60 mJ/m², improving dispersion and impact properties. Coated grades are essential for compounding applications.
Glass Fibers and Glass Beads
Glass fibers are among the most effective fillers for increasing tensile strength and flexural modulus due to their high aspect ratio. In PA6, 30% glass fiber increases tensile strength from 75 MPa to 170 MPa (2.3x) and flexural modulus from 2.7 GPa to 8.5 GPa (3.1x).
However, fiber orientation during molding creates anisotropic shrinkage — lower shrinkage in the flow direction and higher shrinkage perpendicular to flow. This increases warp risk in complex geometries.
Glass beads are symmetric, distribute evenly in the matrix, do not alter shrink directionality, and improve thermal deflection without adding structural strength. Better suited for dimensional stability needs where warp must be minimized.
Talc and Mica
Talc has a platelet structure that improves stiffness, creep resistance, and heat deflection temperature in polypropylene. It nucleates PP crystallization, contributing to higher stiffness, and is standard in automotive interior parts like dashboards and door panels at loading levels of 10%, 15%, or 30% by weight.

Mica, also plate-like but with higher aspect ratio, is used to reduce moisture vapor and oxygen transmission (barrier properties) and improve dimensional stability in electrical components. Plate-like fillers require coupling agents for adequate polymer-filler adhesion.
Wollastonite and Kaolin
Wollastonite has an acicular (needle-like) structure that can partially replace glass fibers. Key properties:
- Improves surface finish alongside tensile strength gains
- High dielectric strength — relevant for electrical applications
- Compatible with coupling agents for better matrix adhesion
Kaolin serves more varied roles across polymer applications:
- Improves anti-blocking performance in film
- Increases impact strength and abrasion resistance
- Functions as an infrared absorber in laser-marked plastics
- Metakaolin grade stabilizes PVC formulations
Nanofillers
Nanofillers (particle size < 100 nm) provide an extremely high surface area-to-volume ratio, enabling significant property improvements at low loading levels — including improved barrier properties, flame retardancy, and scratch resistance.
Main categories:
- Nanoplates (for example, nanoclay — analogous to talc but nanoscale; primary use in barrier and flame retardancy applications)
- Nanofibers (high aspect ratio; improve mechanical strength and stiffness at very low loadings)
- Nanoparticles (isotropic geometry; used for scratch resistance, UV stability, and optical effects)
At 6.4 wt% nanoclay with biaxial stretching, oxygen permeability in PP nanocomposites dropped by 64%. However, barrier improvement disappeared above 3.4 wt% without adequate dispersion due to particle agglomeration.
Poor dispersion negates every performance advantage — and at sufficient agglomeration, it actively weakens the matrix. Achieving exfoliation-level dispersion requires advanced compounding technology, making process control as important as filler selection.
How to Choose the Right Additive or Filler
Match Function to End-Use Requirement
The first selection filter is the performance gap being addressed:
| Performance Need | Solution |
|---|---|
| Thermal degradation during processing | Thermal stabilizer |
| Outdoor UV exposure | HALS + UV absorber |
| Fire safety certification | Flame retardant (halogen-free for wire & cable/E&E) |
| Impact resistance | Fibrous filler (glass/wollastonite) |
| Cost reduction without property sacrifice | CaCO₃ or talc |
| Flexibility in PVC | Plasticizer (40-65 phr for standard flexible) |
| Dimensional stability without warp | Glass beads or spherical filler |
| Barrier properties | Nanoclay or mica (plate-like structure) |
Consider Polymer-Filler Compatibility and Coupling Agents
The polymer-filler interface is critical. A poorly bonded filler acts as a stress concentrator and worsens properties it was meant to improve.
Silane coupling agents are the most widely used for glass and mineral fillers. General structure: (RO)₃-Si-R'-X where alkoxy groups bond to inorganic substrates via Si-O-Metal covalent bonds, and organofunctional groups (amino, epoxy, vinyl) react with the polymer matrix.
Maleic anhydride-grafted polyolefins (MAPP) are used for talc and mineral fillers in polyolefin systems. MAPP functions through dual polarity: non-polar backbone entangles with the PP matrix while anhydride groups react with polar filler surfaces.
Dispersion quality during compounding — screw design, temperature profile, and mixing intensity — determines how well any coupling chemistry actually performs in practice.
Account for Processing Method and Geometry
Different processing methods impose different constraints:
- Injection molding with complex geometry: Glass fiber orientation and anisotropic shrinkage can cause warp. Powder or bead fillers are lower risk.
- Film extrusion: Filler particle size and surface treatment affect optical properties and film integrity. Anti-blocking agents are essential for winding.
- Wire & cable extrusion: Flame retardant mineral fillers like ATH must be compounded at lower temperatures to prevent premature decomposition.
Work with an Experienced Formulation Partner
Additive-filler interactions are complex. Some combinations are synergistic (e.g., flame retardant + synergist packages) while others interfere with each other or with the base resin. Getting the formulation right requires application-specific lab trials, not standardized starting formulations.
Distil's R&D team — drawing on experience from Dow, BASF, L'Oréal, Huntsman, and Reliance — runs application-specific formulation development and managed trial programs. The process covers:
- Problem definition and performance gap analysis
- Formulation design and coupling agent selection
- Validation testing and process optimization
- Scale-up to commercial production via a 20+ partner manufacturing network
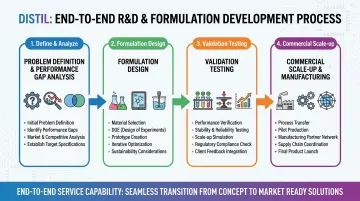
Compounders and brand manufacturers get a single point of contact from lab-scale trials through commercial supply.
Industry-Specific Applications: Where Additives and Fillers Make the Biggest Difference
Automotive and Electrical & Electronics
Automotive plastics face some of the most demanding additive requirements across any industry. Key performance targets include:
- Heat deflection: Talc-filled PP for dashboards and door panels
- UV stability: HALS systems for exterior trims exposed to weathering
- Flame performance: Halogen-free FR systems for EV battery housings requiring UL 94 V-0 certification
The global EV battery housing market was predicted to reach USD 4.47 billion by 2025, reflecting how sharply EV growth is driving FR specification across automotive supply chains.
Electronics present a distinct set of challenges beyond flame retardancy. Circuit boards and enclosures need dimensional stability under thermal cycling, thermal conductivity in heat-sensitive assemblies, and phosphorus-based FR systems rated to UL 94 V-0. That standard requires maximum individual afterflame ≤10 seconds and no flaming drips — a performance level achievable only with properly formulated FR packages.
Wire & Cable and Flexible Packaging
Wire & cable insulation relies on plasticized PVC (flexibility at 40-50 phr for insulation, 50-60 phr for sheathing) combined with halogen-free flame retardants (ATH, MDH) for jacketing compounds meeting fire safety standards like IEC 60332 and EN 50399. The global HFFR market was valued at USD 12.8 billion in 2025 and is projected to reach USD 22.6 billion by 2034.
Flexible packaging — both FMCG and industrial grades — relies on a different set of performance additives:
- Anti-blocking agents: Prevent film layers from sticking during processing and winding
- Slip agents: Control coefficient of friction for high-speed converting lines
- UV stabilizers: Protect contents during outdoor or extended storage
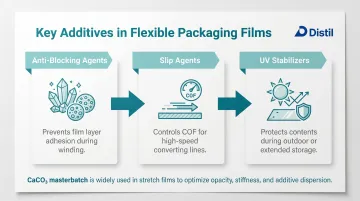
Fillers see limited use in thin films, though CaCO₃-loaded masterbatches appear in stretch films for cost reduction and stiffness control.
Frequently Asked Questions
What are fillers and additives in plastic?
Additives are chemical compounds, typically under 5 wt%, that modify a plastic's processing behavior or end-use properties. Common examples include antioxidants (prevent degradation) and flame retardants (meet fire codes). Fillers are inorganic particles or fibers added in larger volumes (10–65 wt%) to reinforce the material, reduce cost, or impart specific mechanical or thermal properties like stiffness or heat deflection.
What is the difference between a filler and an additive in polymers?
The key distinction is mechanism. Additives work chemically at low concentrations, protecting the polymer or modifying its behavior. Fillers work physically, occupying volume in the polymer matrix to deliver structural reinforcement or property modification at scale.
What are the most common fillers used in plastic manufacturing?
The most widely used fillers are calcium carbonate (cost reduction, stiffness), talc (heat deflection, creep resistance in PP), glass fibers (tensile strength, modulus), kaolin (anti-blocking, impact strength), and carbon black (UV protection, conductivity). Selection depends on the target property profile of the end application.
How do polymer additives improve product performance?
Additives serve three broad functions: protecting the polymer (stabilizers prevent oxidative and UV degradation), enabling safe processing (lubricants reduce melt viscosity; thermal stabilizers prevent chain scission), and meeting regulatory or functional requirements (flame retardants achieve UL 94 ratings; plasticizers create flexible PVC). Each additive type targets a specific performance gap.
What are the 4 types of polymers?
The four broad classifications are thermoplastics (melt-processable, bonded by Van der Waals forces), thermosets (permanently cross-linked during curing, cannot be remelted), elastomers (amorphous, high elastic deformation with light cross-linking), and synthetic fibers (high tensile strength, spun into filaments). Each class carries distinct additive and filler compatibility requirements.
Can multiple additives and fillers be combined in the same polymer formulation?
Yes. Most commercial compounds use multiple components simultaneously — a talc-filled PP automotive part, for instance, typically also contains UV stabilizers, thermal stabilizers, and lubricants. Compatibility must be validated, as some combinations are synergistic (flame retardant + synergist packages) while others interfere, making application-specific trials essential.



