
Introduction
A mid-sized personal care manufacturer receives a customer complaint: a batch of sunscreen has developed discoloration during shelf testing. The root-cause investigation traces the defect to an out-of-spec zinc oxide UV filter containing elevated heavy metal levels. The material passed visual inspection and paperwork review, but no one caught the compositional deviation until the finished product failed.
Raw material quality control (QC) is the first place in your process where a defect can be caught and stopped — before it reaches formulation, batch production, or a customer's shelf. Variability in incoming materials, whether identity mismatch, purity drift, or functional parameter deviation, propagates through every downstream step.
The cost of getting this wrong is measurable. According to the American Society for Quality, poor quality typically accounts for 15–20% of sales revenue in manufacturing operations, with prevention costing a fraction of what failures cost downstream.
This article covers what raw material QC involves, the key steps and testing types, relevant compliance standards, common challenges, and best practices for building a reliable QC program that protects product integrity and brand reputation.
TLDR:
- Raw material QC verifies identity, purity, composition, and functional properties before production use
- Core process spans supplier qualification, sampling, lab testing, release decisions, and traceability
- Testing spans chemical/analytical, physical/functional, and microbiological methods — both compendial and validated in-house
- Compliance frameworks include ISO 9001, cGMP, ISO/IEC 17025, REACH, and pharmacopeial standards
- Lot-to-lot variability, multi-site coordination, and timeline pressure are persistent challenges
What Is Raw Material Quality Control?
Raw material quality control is the systematic process used to verify that incoming materials meet predefined specifications before they enter production. Unlike finished-product QC — which is corrective — raw material QC is preventive. It establishes the quality baseline for everything downstream: formulation accuracy, process reproducibility, batch yield, and product performance.
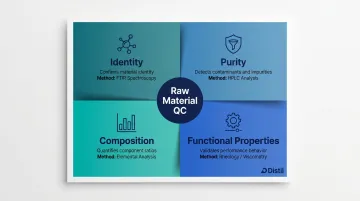
Four core attributes are evaluated:
- Identity: Confirms the material matches its expected chemical or structural profile — typically via FTIR spectroscopy or chromatographic fingerprinting
- Purity: Detects contaminants, residual solvents, heavy metals, or microbial load that could compromise safety or efficacy
- Composition: Quantifies active constituents or polymer characteristics to ensure batch-to-batch uniformity
- Functional properties: Measures physical attributes — particle size, viscosity, moisture content, pH — that directly affect processability and final product behaviour
QA vs. QC: Complementary Roles
Quality Assurance (QA) designs and owns the system: specifications, procedures, supplier qualification protocols, and audit schedules. Quality Control (QC) executes the individual tests and makes release decisions. When QA and QC operate in sync, incoming material decisions are faster, better documented, and defensible under regulatory scrutiny.
Why Raw Material QC Matters
Beyond regulatory compliance, raw material QC delivers tangible business outcomes. A 2024 study analyzing FDA cosmetic recalls from 2011–2023 found that 76.8% of recalls were due to microbial contamination, and 10.2% involved inorganic contaminants such as asbestos — issues often traceable to incoming raw materials02898-6/fulltext). Poor-quality incoming materials trigger product failures, batch rejections, reformulation costs, regulatory non-compliance, and brand damage.
In specialty chemicals — personal care, flavours and fragrances, advanced polymers — even minor variation in a single raw material can alter final product texture, efficacy, or shelf life in ways that are difficult and costly to diagnose after the fact.
The consequences are highly specific. Zinc oxide particle size distribution (nano <100 nm vs. micro 200–1,000 nm) directly affects SPF performance and UV-A protection in sunscreens, according to research presented at IFSCC 2024. A specification deviation invisible to visual inspection can invalidate an entire formulation.
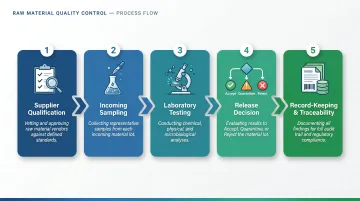
Key Steps in the Raw Material Quality Control Process
Step 1 — Supplier Qualification
Before a material is ordered, establish an Approved Vendor List (AVL). Supplier qualification involves:
- Reviewing documentation: manufacturing licenses, quality certificates, historical Certificate of Analysis (COA) data
- Conducting audits (on-site or remote) to assess manufacturing controls and capability
- Evaluating track record for consistent material delivery
A rigorous AVL directly reduces the rate of incoming material failures at later stages.
Step 2 — Incoming Sampling
Accurate testing depends on representative sampling. Three main approaches:
- Random sampling: Used for homogeneous materials from qualified suppliers
- Stratified sampling: Applied when lots are large or heterogeneous (e.g., bulk powders)
- Risk-based sampling: Calibrates frequency and scope to supplier track record, material criticality, and application risk
Chain-of-custody documentation is critical: every sample must be traceable to supplier, batch number, delivery date, and responsible person. ISO 11648-1:2003 provides statistical guidance for sampling from bulk materials.
Step 3 — Laboratory Testing and Evaluation
Test sampled material against predefined specifications using appropriate analytical methods — compendial or validated non-compendial. Testing scope should be proportionate to risk: a well-known excipient from a qualified supplier warrants fewer tests than a novel material or new source.
Common test categories include:
- Identity testing: Confirms the material is what the supplier declared (IR spectroscopy, TLC, wet chemistry)
- Purity and assay: Quantifies active content and detects impurities or degradation products
- Physical characterisation: Particle size, viscosity, melting point, or other application-critical parameters
- Microbiological testing: Required for materials used in personal care, pharma, or food-contact applications
Step 4 — Release Decision and Documentation
Test results are compared against acceptance criteria. Each lot receives one of three dispositions: accept, quarantine (pending further testing or investigation), or reject. All decisions must be documented with full data traceability under the applicable quality system — cGMP, ISO 9001, or equivalent. Hold materials in a designated quarantine area until a formal release decision is recorded.
The USP General Chapter on incoming material checks notes that identity testing should be performed on every container unless validated exceptions (documented derogations) are in place.
Step 5 — Record-Keeping and Traceability
Retain samples under controlled conditions and maintain batch records for each lot received. These records serve as the audit trail for:
- Regulatory inspections
- Supplier dispute resolution
- Root-cause investigation if downstream product failure occurs
Integrating incoming QC data into a unified quality platform enables real-time tracking across multiple manufacturing sites or supplier partners — critical for businesses operating distributed production networks. Distil's approach across its 20+ manufacturing partner network illustrates this directly: a single quality layer applied consistently from lab to commercial scale eliminates process drift without requiring each partner facility to operate an independent QC infrastructure.
Main Types of Raw Material Quality Testing
Chemical and Analytical Testing
The most common category, covering:
- Identity confirmation: FTIR, NMR
- Purity and composition quantification: HPLC, UV-Vis
- Residual solvent and volatile impurity profiling: GC, GC-MS
- Elemental/heavy metal analysis: ICP-MS, XRF
For fragrance ingredients, GC-MS is used to screen for IFRA allergens (57 suspected allergenic compounds regulated by the International Fragrance Association).
Physical and Functional Testing
Measures properties that determine how a material will behave in processing and in the final product:
- Particle size distribution
- Viscosity
- Moisture content
- Density
- pH
- Thermal behavior (DSC/TGA)
In specialty chemicals, physical attributes are often as critical as chemical identity. For example, zinc oxide particle size directly impacts transparency, SPF efficacy, and UV-A protection in sunscreen formulations.
Microbiological Testing
This testing applies wherever raw materials could carry bioburden, endotoxins, or contamination risk, particularly in personal care, healthcare, and food-contact applications.
Key tests include:
- Bioburden enumeration
- Endotoxin (LAL) testing
- Pathogen screening
ISO 17516:2014 defines microbiological limits for cosmetics. A 2024 study of cosmetic raw materials in Iran found 56.25% contamination rates in untested incoming materials, underscoring the importance of routine microbiological QC.
Choosing the Right Testing Approach
Two decisions shape how testing is structured: whether to follow compendial standards and where the testing happens.
| Dimension | Option A | Option B |
|---|---|---|
| Method type | Compendial (USP, Ph. Eur., JP) — used for pharmaceutical excipients and API-adjacent materials with existing monographs | Non-compendial — validated in-house or by a contract lab for specialty or novel materials without official monographs |
| Testing location | In-house rapid screening (moisture analyzers, FTIR, visual inspection) for fast release decisions | Accredited external labs (ISO/IEC 17025) for complex analyses, regulatory submissions, microbiological testing, and dispute resolution |
The choice of method type depends on regulatory pathway and material class. An effective QC program uses both location approaches in a tiered structure.
Industry Standards and Compliance Frameworks
Core Quality Management Standards
- ISO 9001: Baseline for quality management systems, defining documentation, process control, and continuous improvement requirements
- ISO/IEC 17025: Benchmark for laboratory competence and accreditation — testing methods must be validated and results traceable to recognised standards
- cGMP (current Good Manufacturing Practice): Regulatory floor for pharmaceutical, personal care, and food-grade material testing, enforced by FDA and equivalent agencies globally
Pharmacopeial and Chemical Regulatory References
- USP, Ph. Eur., JP monographs: Define identity, purity, and test methods for pharmaceutical-grade raw materials
- REACH (EU): Requires registration of chemical substances manufactured or imported at 1+ tonne/year, with 10-year record retention
- COSMOS/ISO 16128: Standards for natural and organic personal care ingredients; ISO 16128 defines natural origin index methodology, while COSMOS provides certification
ICH Q7 sections 7.10–7.14 outline raw material requirements for active pharmaceutical ingredients, including supplier qualification (sections 7.30–7.31).
Why Standards Matter Across Multi-Tier Supply Chains
When raw materials cross multiple geographies and pass through multiple processing partners, shared standards are what keep quality expectations consistent. Alignment with ISO, cGMP, and pharmacopeial frameworks delivers three practical outcomes:
- Audit defensibility across regulatory jurisdictions (FDA, EMA, BIS)
- Transferable quality criteria when onboarding or switching suppliers
- Traceable documentation that survives supply chain disruptions
Common Challenges in Raw Material Quality Control
Lot-to-Lot Variability
Natural raw materials — botanical extracts, bio-based polymers, fragrance ingredients — inherently vary by source geography, harvest season, or processing method. This variability can push functional attributes outside specification even when chemical identity is confirmed.
For example, lavender essential oil contains over 170 compounds, with linalool typically accounting for 31.1% of composition. Geographic origin, harvest timing, and distillation method all influence chemical profile, creating formulation consistency challenges in personal care and fragrance applications.
Managing Quality Across Multi-Supplier and Multi-Site Networks
When sourcing from multiple approved vendors or operating across distributed manufacturing partners, maintaining specification alignment and consistent test execution is difficult to manage consistently. Common failure points include:
- Inconsistent sampling procedures across sites producing non-comparable data
- Different lab instruments or calibration standards yielding conflicting results for the same material
- Varying analyst interpretation of borderline results, leading to inconsistent release decisions

Timeline Pressure Versus Testing Rigor
Production schedules create pressure to release incoming materials quickly. Compressed timelines increase the risk of incomplete testing, inadequate documentation, or premature release of borderline-compliant material. The downstream cost — production delays, batch failures, rework — exceeds the time saved by shortcutting incoming QC.
Best Practices for an Effective Raw Material QC Program
Apply Risk-Based Sampling
Not every incoming lot requires the same depth of testing. Calibrate testing frequency and scope to:
- Supplier track record
- Material criticality
- Application risk
- Historical COA consistency
New suppliers or materials entering high-risk applications warrant full testing of every lot. Established partners with consistent performance can qualify for reduced-frequency or COA-release protocols — with defined trigger conditions for escalation.
Define Material Specifications Before Sourcing
Clear, measurable acceptance criteria for every critical quality attribute (identity, purity, physical parameters) should be established collaboratively by R&D, QA, and procurement before a supplier is selected. Specifications developed after problems arise are reactive and harder to enforce contractually.
Build a Unified Quality Data Layer
Connect incoming test results, supplier COA data, batch records, and production performance into a single system visible across QA, procurement, and operations. This shifts raw material QC from a gatekeeping function into an ongoing supplier intelligence tool — enabling early identification of drift or degrading performance before a non-conformance occurs.
This approach is especially relevant for asset-light, multi-site models. Distil, for example, applies a unified quality system across its 20+ manufacturing partner network, embedding quality infrastructure at every production node without requiring centralised capital assets.
Frequently Asked Questions
What is quality control of raw materials?
Raw material quality control verifies that incoming materials meet predefined specifications for identity, purity, composition, and functional properties before production use. This ensures downstream manufacturing starts from a controlled, consistent base.
What are the key steps in raw material quality control?
Five core steps govern the process: supplier qualification (Approved Vendor List), representative incoming sampling with chain-of-custody documentation, laboratory testing against specifications, a documented release decision (accept/quarantine/reject), and traceability through record-keeping and retained samples.
What are the main types of quality control for raw materials?
Three testing categories cover most materials: chemical/analytical (identity and purity via FTIR, HPLC, GC-MS), physical/functional (viscosity, particle size, moisture), and microbiological (bioburden, endotoxin, pathogens). Method selection — compendial or validated non-compendial — depends on the material type and applicable regulatory requirement.
What are the responsibilities of raw material quality control?
Responsibilities are shared across three functions. QA owns specifications, procedures, and the supplier qualification framework. QC executes testing and makes release decisions. Procurement enforces documentation requirements and manages COA compliance with suppliers.
What are the common challenges in raw material quality control?
Natural lot-to-lot variability — particularly in botanicals and bio-based ingredients — is the most persistent challenge. Beyond variability, maintaining consistent QC standards across multi-supplier or multi-site networks is difficult to govern. A third pressure point is the recurring tension between production timelines and the thoroughness required for defensible testing.
How does raw material quality control affect product performance?
Raw material attributes directly determine finished product behavior. Variations in purity, particle size, moisture, or functional group chemistry can alter formulation efficacy, texture, stability, or shelf life — making incoming QC the single most impactful intervention point for product consistency.



